Seasonal variation in the balance and strength of cooperative and competitive behavior in patches of blue mussels
- PMID: 37856481
- PMCID: PMC10586602
- DOI: 10.1371/journal.pone.0293142
Seasonal variation in the balance and strength of cooperative and competitive behavior in patches of blue mussels
Abstract
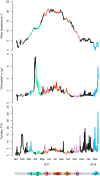
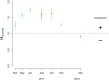
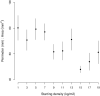
Aggregation into groups may affect performance of individuals through the balance and strength of facilitative versus competitive interactions. We studied in situ how seasonal variation in abiotic environment affects this balance for blue mussels, a semi-sessile species. We hypothesize that seasonal variation in stresses and resources affects the strength of the interaction. We expected that, in benign conditions (here: high food availability, medium temperatures, low hydrodynamic stress), performance is dominated by growth and is better at low densities, while at adverse conditions (here: low food availability, low or high temperatures, high hydrodynamic stress), performance is dominated by survival and higher at high densities. Mussels were kept in shallow subtidal exclosures at 10 different densities for a one-month period. This exact procedure was repeated seven times at the same location within a one-year period. We measured development in mussel patch shape, performance, and environmental parameters. Environmental conditions for mussels were most benign in summer and most adverse in winter. Patches developed into less complex shapes at lower densities, but also after stronger hydrodynamic disturbances. Towards summer, mussels became more active, aggregation behavior increased, and interactions became more pronounced. Towards winter, mussels became less active: aggregation behavior and growth rates declined and at the lowest temperatures survival started to decrease with mussel density. Survival and growth (by proxy of mussel condition) were both density-dependent; however, contrary to our expectations we found positive interactions between density and survival at the most benign conditions in summer and negative interactions at the most adverse conditions in winter. In between the two seasons, the strength of the interactions increased towards summer and decreased towards winter following a bell-shaped pattern. This pattern might be explained by the environmental mediated aggregation behavior of the mussels. The obvious seasonal pattern in balance and strength of density-dependent interactions demonstrates that strength and direction of intra-specific interactions are both strongly affected by environmental context.
Copyright: © 2023 Capelle et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Krause J, Ruxton GD, Ruxton G, Ruxton IG. Living in groups: Oxford University Press; 2002.
-
- Turchin P, Kareiva P. Aggregation in Aphis varians: an effective strategy for reducing predation risk. Ecology. 1989;70(4):1008–16.
-
- Connell SD. Is there safety‐in‐numbers for prey? Oikos. 2000;88(3):527–32.
-
- Cocroft RB. Vibrational communication and the ecology of group-living, herbivorous insects. American Zoologist. 2001;41(5):1215–21.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

