Ultradian hydrocortisone replacement alters neuronal processing, emotional ambiguity, affect and fatigue in adrenal insufficiency: The PULSES trial
- PMID: 37857352
- PMCID: PMC10952319
- DOI: 10.1111/joim.13721
Ultradian hydrocortisone replacement alters neuronal processing, emotional ambiguity, affect and fatigue in adrenal insufficiency: The PULSES trial
Erratum in
-
Correction to "Ultradian hydrocortisone replacement alters neuronal processing, emotional ambiguity, affect and fatigue in adrenal insufficiency: The PULSES trial".J Intern Med. 2025 Apr;297(4):452. doi: 10.1111/joim.20076. J Intern Med. 2025. PMID: 40097908 Free PMC article. No abstract available.
Abstract
Background: Primary adrenal insufficiency (PAI) mortality and morbidity remain unacceptably high, possibly arising as glucocorticoid replacement does not replicate natural physiology. A pulsatile subcutaneous pump can closely replicate cortisol's circadian and ultradian rhythm.
Objectives: To assess the effect of pump therapy on quality of life, mood, functional neuroimaging, behavioural/cognitive responses, sleep and metabolism.
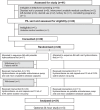
Methods: A 6-week randomised, crossover, double-blinded and placebo-controlled feasibility study of usual dose hydrocortisone in PAI administered as either pulsed subcutaneous or standard care in Bristol, United Kingdom (ISRCTN67193733). Participants were stratified by adrenal insufficiency type. All participants who received study drugs are included in the analysis. The primary outcome, the facial expression recognition task (FERT), occurred at week 6.
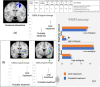
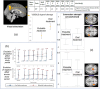
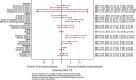
Results: Between December 2014 and 2017, 22 participants were recruited - 20 completed both arms, and 21 were analysed. The pump was well-tolerated. No change was seen in the FERT primary outcome; however, there were subjective improvements in fatigue and mood. Additionally, functional magnetic resonance imaging revealed differential neural processing to emotional cues and visual stimulation. Region of interest analysis identified the left amygdala and insula, key glucocorticoid-sensitive regions involved in emotional ambiguity. FERT post hoc analysis confirmed this response. There were four serious adverse events (AE): three intercurrent illnesses requiring hospitalisation (1/3, 33.3% pump) and a planned procedure (1/1, 100% pump). There was a small number of expected AEs: infusion site bruising/itching (3/5, 60% pump), intercurrent illness requiring extra (3/7, 42% pump) and no extra (4/6, 66% pump) steroid.
Conclusions: These findings support the administration of hormone therapy that mimics physiology.
Keywords: fMRI; glucocorticoid replacement therapy; primary adrenal insufficiency; ultradian.
© 2023 The Authors. Journal of Internal Medicine published by John Wiley & Sons Ltd on behalf of Association for Publication of The Journal of Internal Medicine.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures

References
-
- Chabre O, Goichot B, Zenaty D, Bertherat J. Group 1. Epidemiology of primary and secondary adrenal insufficiency: prevalence and incidence, acute adrenal insufficiency, long‐term morbidity and mortality. Ann Endocrinol (Paris). 2017;78:490–494. - PubMed
-
- Erichsen MM, LøvåS K, Skinningsrud B, Wolff AB, Undlien DE, Svartberg J, et al. Clinical, immunological, and genetic features of autoimmune primary adrenal insufficiency: observations from a Norwegian registry. J Clin Endocrinol Metab. 2009;94:4882–4890. - PubMed
-
- Johannsson G, Falorni A, Skrtic S, Lennernäs H, Quinkler M, Monson JP, et al. Adrenal insufficiency: review of clinical outcomes with current glucocorticoid replacement therapy. Clin Endocrinol (Oxf). 2015;82:2–11. - PubMed
-
- Øksnes M, Bensing S, Hulting A‐L, Kämpe O, Hackemann A, Meyer G, et al. Quality of life in European patients with Addison's disease: validity of the disease‐specific questionnaire AddiQoL. J Clin Endocrinol Metab. 2012;97:568–576. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

