Clinical and regulatory development strategies for Shigella vaccines intended for children younger than 5 years in low-income and middle-income countries
- PMID: 37858591
- PMCID: PMC10603611
- DOI: 10.1016/S2214-109X(23)00421-7
Clinical and regulatory development strategies for Shigella vaccines intended for children younger than 5 years in low-income and middle-income countries
Abstract
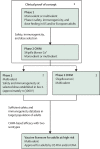
Shigellosis causes considerable public health burden, leading to excess deaths as well as acute and chronic consequences, particularly among children living in low-income and middle-income countries (LMICs). Several Shigella vaccine candidates are advancing in clinical trials and offer promise. Although multiple target populations might benefit from a Shigella vaccine, the primary strategic goal of WHO is to accelerate the development and accessibility of safe, effective, and affordable Shigella vaccines that reduce mortality and morbidity in children younger than 5 years living in LMICs. WHO consulted with regulators and policy makers at national, regional, and global levels to evaluate pathways that could accelerate regulatory approval in this priority population. Special consideration was given to surrogate efficacy biomarkers, the role of controlled human infection models, and the establishment of correlates of protection. A field efficacy study in children younger than 5 years in LMICs is needed to ensure introduction in this priority population.
Copyright © 2023 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests NB provides regulatory advice to the regulated industry. PBP receives funding from the Bill & Melinda Gates Foundation for Enterics for Global Health Shigella surveillance. DCK received funding from the Bill & Melinda Gates Foundation while at PATH, and chaired the WHO Product Development for Vaccines Advisory Committee until 2022. MSR consulted for Emergent Biosolutions and Limmatech Biologics while at the University of Nevada. All other authors declare no competing interests.
Figures

References
-
- Institute for Health Metrics and Evaluation 2019 Global Burden of Disease study. 2019. https://vizhub.healthdata.org/gbd-results
-
- Kotloff KL, Riddle MS, Platts-Mills JA, Pavlinac P, Zaidi AKM. Shigellosis. Lancet. 2018;391:801–812. - PubMed
-
- WHO WHO preferred product characteristics for vaccines against Shigella. 2021. https://www.who.int/publications/i/item/9789240036741
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

