Single-cell transcriptomes reveal the heterogeneity and microenvironment of vestibular schwannoma
- PMID: 37862593
- PMCID: PMC10912001
- DOI: 10.1093/neuonc/noad201
Single-cell transcriptomes reveal the heterogeneity and microenvironment of vestibular schwannoma
Abstract
Background: Vestibular schwannoma (VS) is the most common benign tumor in the cerebellopontine angle and internal auditory canal. Illustrating the heterogeneous cellular components of VS could provide insights into its various growth patterns.
Methods: Single-cell RNA sequencing was used to profile transcriptomes from 7 VS samples and 2 normal nerves. Multiplex immunofluorescence was employed to verify the data set results. Bulk RNA sequencing was conducted on 5 normal nerves and 44 VS samples to generate a prediction model for VS growth.
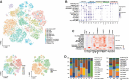
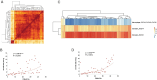
Results: A total of 83 611 cells were annotated as 14 distinct cell types. We uncovered the heterogeneity in distinct VS tumors. A subset of Schwann cells with the vascular endothelial growth factor biomarker was significantly associated with fast VS growth through mRNA catabolism and peptide biosynthesis. The macrophages in the normal nerves were largely of the M2 phenotype, while no significant differences in the proportions of M1 and M2 macrophages were found between slow-growing and fast-growing VS. The normal spatial distribution of fibroblasts and vascular cells was destroyed in VS. The communications between Schwann cells and vascular cells were strengthened in VS compared with those in the normal nerve. Three cell clusters were significantly associated with fast VS growth and could refine the growth classification in bulk RNA.
Conclusions: Our findings offer novel insights into the VS microenvironment at the single-cell level. It may enhance our understanding of the different clinical phenotypes of VS and help predict growth characteristics. Molecular subtypes should be included in the treatment considerations.
Keywords: heterogeneity; molecular subtypes; single-cell RNA sequencing; tumor microenvironment; vestibular schwannoma (VS).
© The Author(s) 2023. Published by Oxford University Press on behalf of the Society for Neuro-Oncology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
None declared.
Figures




Similar articles
-
Single-Cell RNA-Seq Reveals Heterogeneity of Cell Communications between Schwann Cells and Fibroblasts within Vestibular Schwannoma Microenvironment.Am J Pathol. 2022 Sep;192(9):1230-1249. doi: 10.1016/j.ajpath.2022.06.006. Epub 2022 Jun 22. Am J Pathol. 2022. PMID: 35750260
-
Immunohistochemical profile of cytokines and growth factors expressed in vestibular schwannoma and in normal vestibular nerve tissue.Mol Med Rep. 2015 Jul;12(1):737-45. doi: 10.3892/mmr.2015.3415. Epub 2015 Mar 4. Mol Med Rep. 2015. PMID: 25738867
-
Interplay between VEGF-A and cMET signaling in human vestibular schwannomas and schwann cells.Cancer Biol Ther. 2015;16(1):170-5. doi: 10.4161/15384047.2014.972765. Cancer Biol Ther. 2015. PMID: 25692621 Free PMC article.
-
[Neurotrophic factor expression in vestibular schwannoma. An overview].Laryngorhinootologie. 2006 Oct;85(10):731-7. doi: 10.1055/s-2006-925286. Epub 2006 Apr 10. Laryngorhinootologie. 2006. PMID: 16612755 Review. German.
-
Tumor Biology and Microenvironment of Vestibular Schwannoma-Relation to Tumor Growth and Hearing Loss.Biomedicines. 2022 Dec 23;11(1):32. doi: 10.3390/biomedicines11010032. Biomedicines. 2022. PMID: 36672540 Free PMC article. Review.
Cited by
-
Multiplatform molecular analyses reveal two molecular subgroups of NF2-related schwannomatosis vestibular schwannomas with distinct tumour microenvironment and therapeutic vulnerabilities.Acta Neuropathol. 2025 May 9;149(1):47. doi: 10.1007/s00401-025-02883-6. Acta Neuropathol. 2025. PMID: 40343504
-
SMARCB1-related schwannomatosis and other SMARCB1-associated phenotypes: clinical spectrum and molecular pathogenesis.Fam Cancer. 2025 Aug 12;24(3):64. doi: 10.1007/s10689-025-00486-4. Fam Cancer. 2025. PMID: 40794298 Free PMC article. Review.
-
Development of a 23-Gene Signature for Tumor Growth Mechanism in Vestibular Schwannoma.Cancers (Basel). 2024 Dec 11;16(24):4134. doi: 10.3390/cancers16244134. Cancers (Basel). 2024. PMID: 39766034 Free PMC article.
-
Integrating single-cell and spatial transcriptomics reveals the cellular heterogeneity of vestibular schwannoma.NPJ Precis Oncol. 2025 Jul 8;9(1):228. doi: 10.1038/s41698-025-01028-y. NPJ Precis Oncol. 2025. PMID: 40629113 Free PMC article.
-
A single-cell atlas of Schwannoma across genetic backgrounds and anatomic locations.Genome Med. 2025 Apr 11;17(1):37. doi: 10.1186/s13073-025-01462-4. Genome Med. 2025. PMID: 40217315 Free PMC article.
References
-
- Evans DG, Moran A, King A, et al. . Incidence of vestibular schwannoma and neurofibromatosis 2 in the North West of England over a 10-year period: higher incidence than previously thought. Otol Neurotol. 2005;26(1):93–97. - PubMed
-
- Carlson ML, Link MJ.. Vestibular schwannomas. N Engl J Med. 2021;384(14):1335–1348. - PubMed
-
- Charabi S, Mantoni M, Tos M, Thomsen J.. Cystic vestibular schwannomas: neuroimaging and growth rate. J Laryngol Otol. 1994;108(5):375–379. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
- 81900928/National Natural Science Foundation of China
- 20191913/Shanghai Jiao Tong University
- YBKB201902/Biobank Project of Shanghai Ninth People's Hospital
- 2018SHZDZK05/Shanghai Municipal Science and Technology Major Project
- 14DZ2260300/Shanghai Key Laboratory of Translational Medicine on Ear and Nose Diseases
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

