Advax-CpG55.2-adjuvanted monovalent or trivalent SARS-CoV-2 recombinant spike protein vaccine protects hamsters against heterologous infection with Beta or Delta variants
- PMID: 37863669
- PMCID: PMC10873063
- DOI: 10.1016/j.vaccine.2023.10.018
Advax-CpG55.2-adjuvanted monovalent or trivalent SARS-CoV-2 recombinant spike protein vaccine protects hamsters against heterologous infection with Beta or Delta variants
Abstract
The ongoing evolution of SARS-CoV-2 variants emphasizes the need for vaccines providing broad cross-protective immunity. This study was undertaken to assess the ability of Advax-CpG55.2 adjuvanted monovalent recombinant spike protein (Wuhan, Beta, Gamma) vaccines or a trivalent formulation to protect hamsters againstBeta or Delta virus infection. The ability of vaccines to block virus transmission to naïve co-housed animals was also assessed. In naïve hosts, the Beta variant induced higher virus loads than the Delta variant, and conversely the Delta variant caused more severe disease and was more likely to be associated with virus transmission. The trivalent vaccine formulation provided the best protection against both Beta and Delta infection and also completely prevented virus transmission. The next best performing vaccine was the original monovalent Wuhan-based vaccine. Notably, hamsters that received the monovalent Gamma spike vaccine had the highest viral loads and clinical disease of all the vaccine groups, a potential signal of antibody dependent-enhancement (ADE). These hamsters were also the most likely to transmit Delta virus to naïve recipients. In murine studies, the Gamma spike vaccine induced the highest total spike protein to RBD IgG ratio and the lowest levels of neutralizing antibody, a context that could predispose to ADE. Overall, the study results confirmed that the current SpikoGen® vaccine based on Wuhan spike protein was still able to protect against clinical disease caused by either the Beta or Delta virus variants but suggested additional protection may be obtained by combining it with extra variant spike proteins to make a multivalent formulation. This study highlights the complexity of optimizing vaccine protection against multiple SARS-CoV-2 variants and stresses the need to continue to pursue new and improved COVID-19 vaccines able to provide robust, long-lasting, and broadly cross-protective immunity against constantly evolving SARS-CoV-2 variants.
Keywords: Adjuvant; Advax; COVID-19; Coronavirus; Pandemic; SARS-Cov-2; Vaccine.
Copyright © 2023 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: YHO and NP are affiliated with Vaxine Pty Ltd which holds rights to COVAX-19/Spikogen vaccine and Advax™ and CpG55.2™ adjuvants.
Figures

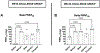




Similar articles
-
Clinical development of SpikoGen®, an Advax-CpG55.2 adjuvanted recombinant spike protein vaccine.Hum Vaccin Immunother. 2024 Dec 31;20(1):2363016. doi: 10.1080/21645515.2024.2363016. Epub 2024 Jun 5. Hum Vaccin Immunother. 2024. PMID: 38839044 Free PMC article. Review.
-
Study of immunogenicity and efficacy against Omicron BA.5 of recombinant protein-based COVID-19 vaccine delivered by intramuscular and mucosal routes in nonhuman primates.Vaccine. 2024 Feb 15;42(5):1122-1135. doi: 10.1016/j.vaccine.2024.01.034. Epub 2024 Jan 22. Vaccine. 2024. PMID: 38262808
-
Immunogenicity and safety study of a single dose of SpikoGen® vaccine as a heterologous or homologous intramuscular booster following a primary course of mRNA, adenoviral vector or recombinant protein COVID-19 vaccine in ambulatory adults.Vaccine. 2025 Mar 7;49:126744. doi: 10.1016/j.vaccine.2025.126744. Epub 2025 Feb 5. Vaccine. 2025. PMID: 39914274 Clinical Trial.
-
Safety and immunogenicity of a modified mRNA-lipid nanoparticle vaccine candidate against COVID-19: Results from a phase 1, dose-escalation study.Hum Vaccin Immunother. 2024 Dec 31;20(1):2408863. doi: 10.1080/21645515.2024.2408863. Epub 2024 Oct 18. Hum Vaccin Immunother. 2024. PMID: 39422261 Free PMC article. Clinical Trial.
-
Antibody tests for identification of current and past infection with SARS-CoV-2.Cochrane Database Syst Rev. 2022 Nov 17;11(11):CD013652. doi: 10.1002/14651858.CD013652.pub2. Cochrane Database Syst Rev. 2022. PMID: 36394900 Free PMC article.
Cited by
-
Effects of age and gender on immunogenicity and reactogenicity of SpikoGen recombinant spike protein vaccine: a post-hoc analysis.Sci Rep. 2024 Sep 30;14(1):22631. doi: 10.1038/s41598-024-67945-3. Sci Rep. 2024. PMID: 39349494 Free PMC article.
-
Clinical development of SpikoGen®, an Advax-CpG55.2 adjuvanted recombinant spike protein vaccine.Hum Vaccin Immunother. 2024 Dec 31;20(1):2363016. doi: 10.1080/21645515.2024.2363016. Epub 2024 Jun 5. Hum Vaccin Immunother. 2024. PMID: 38839044 Free PMC article. Review.
-
Post-Hoc Analysis of Potential Correlates of Protection of a Recombinant SARS-CoV-2 Spike Protein Extracellular Domain Vaccine Formulated with Advax-CpG55.2-Adjuvant.Int J Mol Sci. 2024 Aug 30;25(17):9459. doi: 10.3390/ijms25179459. Int J Mol Sci. 2024. PMID: 39273405 Free PMC article. Clinical Trial.
-
Multivalent and Sequential Heterologous Spike Protein Vaccinations Effectively Induce Protective Humoral Immunity against SARS-CoV-2 Variants.Vaccines (Basel). 2024 Mar 27;12(4):362. doi: 10.3390/vaccines12040362. Vaccines (Basel). 2024. PMID: 38675744 Free PMC article.
References
-
- Clark T, Cassidy-Hanley D. Recombinant subunit vaccines: potentials and constraints. Developments in biologicals. 2005;121:153–63. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

