BDNF-loaded PDADMAC-heparin multilayers: a novel approach for neuroblastoma cell study
- PMID: 37864014
- PMCID: PMC10589271
- DOI: 10.1038/s41598-023-45045-y
BDNF-loaded PDADMAC-heparin multilayers: a novel approach for neuroblastoma cell study
Abstract
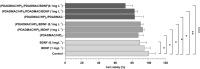
Biomaterial science has contributed tremendously to developing nanoscale materials for delivering biologically active compounds, enhancing protein stability, and enabling its therapeutic use. This paper presents a process of formation of polyelectrolyte multilayer (PEM) prepared by sequential adsorption of positively charged polydiallyldimethylammonium chloride (PDADMAC) and negatively charged heparin sodium salt (HP), from low polyelectrolyte concentration, on a solid substrate. PEM was further applied as a platform for the adsorption of a brain-derived growth factor (BDNF), which is a protein capable of regulating neuronal cell development. The multilayers containing BDNF were thoroughly characterized by electrokinetic (streaming potential measurements, SPM) and optical (optical waveguide lightmode spectroscopy, OWLS) techniques. It was found that BDNF was significantly adsorbed onto polyelectrolyte multilayers terminated by HP under physiological conditions. We further explore the effect of established PEMs in vitro on the neuroblastoma SH-SY5Y cell line. An enzyme-linked immunosorbent assay (ELISA) confirmed that BDNF was released from multilayers, and the use of the PEMs intensified its cellular uptake. Compared to the control, PEMs with adsorbed BDNF significantly reduced cell viability and mitochondrial membrane polarization to as low as 72% and 58%, respectively. HPLC analysis showed that both PDADMAC-terminated and HP-terminated multilayers have antioxidative properties as they almost by half decreased lipid peroxidation in SH-SY5Y cells. Finally, enhanced formation of spheroid-like, 3D structures was observed by light microscopy. We offer a well-characterized PEM with antioxidant properties acting as a BDNF carrier, stabilizing BDNF and making it more accessible to cells in an inhomogeneous, dynamic, and transient in vitro environment. Described multilayers can be utilized in future biomedical applications, such as boosting the effect of treatment by selective anticancer as adjuvant therapy, and in biomedical research for future development of more precise neurodegenerative disease models, as they enhance cellular 3D structure formation.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors declare no competing interests.
Figures








References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

