Lipocalin 2 regulates mitochondrial phospholipidome remodeling, dynamics, and function in brown adipose tissue in male mice
- PMID: 37872178
- PMCID: PMC10593768
- DOI: 10.1038/s41467-023-42473-2
Lipocalin 2 regulates mitochondrial phospholipidome remodeling, dynamics, and function in brown adipose tissue in male mice
Erratum in
-
Author Correction: Lipocalin 2 regulates mitochondrial phospholipidome remodeling, dynamics, and function in brown adipose tissue in male mice.Nat Commun. 2023 Dec 12;14(1):8231. doi: 10.1038/s41467-023-44147-5. Nat Commun. 2023. PMID: 38086875 Free PMC article. No abstract available.
Abstract
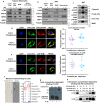
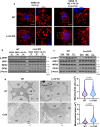
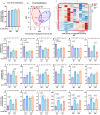
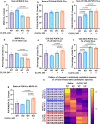
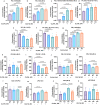
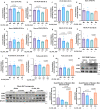
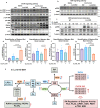
Mitochondrial function is vital for energy metabolism in thermogenic adipocytes. Impaired mitochondrial bioenergetics in brown adipocytes are linked to disrupted thermogenesis and energy balance in obesity and aging. Phospholipid cardiolipin (CL) and phosphatidic acid (PA) jointly regulate mitochondrial membrane architecture and dynamics, with mitochondria-associated endoplasmic reticulum membranes (MAMs) serving as the platform for phospholipid biosynthesis and metabolism. However, little is known about the regulators of MAM phospholipid metabolism and their connection to mitochondrial function. We discover that LCN2 is a PA binding protein recruited to the MAM during inflammation and metabolic stimulation. Lcn2 deficiency disrupts mitochondrial fusion-fission balance and alters the acyl-chain composition of mitochondrial phospholipids in brown adipose tissue (BAT) of male mice. Lcn2 KO male mice exhibit an increase in the levels of CLs containing long-chain polyunsaturated fatty acids (LC-PUFA), a decrease in CLs containing monounsaturated fatty acids, resulting in mitochondrial dysfunction. This dysfunction triggers compensatory activation of peroxisomal function and the biosynthesis of LC-PUFA-containing plasmalogens in BAT. Additionally, Lcn2 deficiency alters PA production, correlating with changes in PA-regulated phospholipid-metabolizing enzymes and the mTOR signaling pathway. In conclusion, LCN2 plays a critical role in the acyl-chain remodeling of phospholipids and mitochondrial bioenergetics by regulating PA production and its function in activating signaling pathways.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Lipocalin 2 regulates brown fat activation via a nonadrenergic activation mechanism.J Biol Chem. 2014 Aug 8;289(32):22063-77. doi: 10.1074/jbc.M114.559104. Epub 2014 Jun 10. J Biol Chem. 2014. PMID: 24917675 Free PMC article.
-
Lipocalin 2, a Regulator of Retinoid Homeostasis and Retinoid-mediated Thermogenic Activation in Adipose Tissue.J Biol Chem. 2016 May 20;291(21):11216-29. doi: 10.1074/jbc.M115.711556. Epub 2016 Mar 22. J Biol Chem. 2016. PMID: 27008859 Free PMC article.
-
Loss of APOO (MIC26) aggravates obesity-related whitening of brown adipose tissue via PPARα-mediated functional interplay between mitochondria and peroxisomes.Metabolism. 2023 Jul;144:155564. doi: 10.1016/j.metabol.2023.155564. Epub 2023 Apr 23. Metabolism. 2023. PMID: 37088120
-
Origins and early development of the concept that brown adipose tissue thermogenesis is linked to energy balance and obesity.Biochimie. 2017 Mar;134:62-70. doi: 10.1016/j.biochi.2016.09.007. Epub 2016 Sep 10. Biochimie. 2017. PMID: 27621146 Review.
-
The Role of Phospholipid Alterations in Mitochondrial and Brain Dysfunction after Cardiac Arrest.Int J Mol Sci. 2024 Apr 24;25(9):4645. doi: 10.3390/ijms25094645. Int J Mol Sci. 2024. PMID: 38731864 Free PMC article. Review.
Cited by
-
m6Am Methyltransferase PCIF1 Promotes LPP3 Mediated Phosphatidic Acid Metabolism and Renal Cell Carcinoma Progression.Adv Sci (Weinh). 2024 Dec;11(46):e2404033. doi: 10.1002/advs.202404033. Epub 2024 Oct 18. Adv Sci (Weinh). 2024. PMID: 39422663 Free PMC article.
-
Phosphatidate phosphatase Lipin1 alters mitochondria-associated endoplasmic reticulum membranes (MAMs) homeostasis: effects which contribute to the development of diabetic encephalopathy.J Neuroinflammation. 2025 Apr 18;22(1):111. doi: 10.1186/s12974-025-03441-3. J Neuroinflammation. 2025. PMID: 40251630 Free PMC article.
-
Exercise Alleviates Aging of Adipose Tissue through Adipokine Regulation.Metabolites. 2024 Feb 22;14(3):135. doi: 10.3390/metabo14030135. Metabolites. 2024. PMID: 38535295 Free PMC article. Review.
-
The Role of Exerkines in Obesity-Induced Disruption of Mitochondrial Homeostasis in Thermogenic Fat.Metabolites. 2024 May 17;14(5):287. doi: 10.3390/metabo14050287. Metabolites. 2024. PMID: 38786764 Free PMC article. Review.
-
Brown adipose tissue: a potential target for aging interventions and healthy longevity.Biogerontology. 2024 Nov;25(6):1011-1024. doi: 10.1007/s10522-024-10137-3. Epub 2024 Oct 8. Biogerontology. 2024. PMID: 39377866 Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

