Evaluating blood oxygen saturation measurements by popular fitness trackers in postoperative patients: A prospective clinical trial
- PMID: 37876822
- PMCID: PMC10590865
- DOI: 10.1016/j.isci.2023.108155
Evaluating blood oxygen saturation measurements by popular fitness trackers in postoperative patients: A prospective clinical trial
Abstract
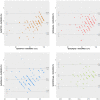
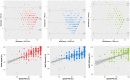
Blood oxygen saturation is an important clinical parameter, especially in postoperative hospitalized patients, monitored in clinical practice by arterial blood gas (ABG) and/or pulse oximetry that both are not suitable for a long-term continuous monitoring of patients during the entire hospital stay, or beyond. Technological advances developed recently for consumer-grade fitness trackers could-at least in theory-help to fill in this gap, but benchmarks on the applicability and accuracy of these technologies in hospitalized patients are currently lacking. We therefore conducted at the postanaesthesia care unit under controlled settings a prospective clinical trial with 201 patients, comparing in total >1,000 oxygen blood saturation measurements by fitness trackers of three brands with the ABG gold standard and with pulse oximetry. Our results suggest that, despite of an overall still tolerable measuring accuracy, comparatively high dropout rates severely limit the possibilities of employing fitness trackers, particularly during the immediate postoperative period of hospitalized patients.
Keywords: Bioelectronics; Clinical measurement in health technology; Health sciences.
© 2023 The Authors.
Conflict of interest statement
S.H., P.R., R.L., B.E.W., M.H., R.P., and M.S. declare no conflicts of interest. P.H. received a research award from Vogel-Foundation and is a member of the Clinician Scientist Program, Wuerzburg. P.M. received honoraria for scientific lectures from CSL Behring GmbH, Haemonetics, Werfen GmbH, and ViforPharma GmbH. P.K. received lecturing fees from TEVA, Sintetica, CSL Behring GmbH, Vifor Pharma GmbH, Pharmacosmos, and Grünenthal and consulted for TEVA and Milestone Scientific Inc. All mentioned funders and especially the manufacturers of the investigated devices had no role in the design of the study; collection, analyses, or interpretation of data; writing of the manuscript; or in the decision to publish the results.
Figures

References
-
- Buekers J., De Boever P., Vaes A.W., Aerts J.-M., Wouters E.F.M., Spruit M.A., Theunis J. Oxygen saturation measurements in telemonitoring of patients with COPD: a systematic review. Expet Rev. Respir. Med. 2018;12:113–123. - PubMed
-
- Buekers J., Theunis J., De Boever P., Vaes A.W., Koopman M., Janssen E.V., Wouters E.F., Spruit M.A., Aerts J.-M. Wearable Finger Pulse Oximetry for Continuous Oxygen Saturation Measurements During Daily Home Routines of Patients With Chronic Obstructive Pulmonary Disease (COPD) Over One Week: Observational Study. JMIR Mhealth Uhealth. 2019;7 - PMC - PubMed
-
- Blackburn J.P. What is new in blood-gas analysis? Br. J. Anaesth. 1978;50:51–62. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

