Gene Expression Within a Human Choroidal Neovascular Membrane Using Spatial Transcriptomics
- PMID: 37878301
- PMCID: PMC10615143
- DOI: 10.1167/iovs.64.13.40
Gene Expression Within a Human Choroidal Neovascular Membrane Using Spatial Transcriptomics
Abstract
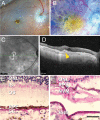
Purpose: Macular neovascularization is a relatively common and potentially visually devastating complication of age-related macular degeneration. In macular neovascularization, pathologic angiogenesis can originate from either the choroid or the retina, but we have limited understanding of how different cell types become dysregulated in this dynamic process.
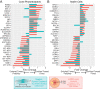
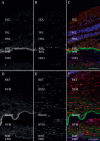
Methods: To study how gene expression is altered in focal areas of pathology, we performed spatial RNA sequencing on a human donor eye with macular neovascularization as well as a healthy control donor. We performed differential expression to identify genes enriched within the area of macular neovascularization and used deconvolution algorithms to predict the originating cell type of these dysregulated genes.
Results: Within the area of neovascularization, endothelial cells demonstrated increased expression of genes related to Rho family GTPase signaling and integrin signaling. Likewise, VEGF and TGFB1 were identified as potential upstream regulators that could drive the observed gene expression changes produced by endothelial and retinal pigment epithelium cells in the macular neovascularization donor. These spatial gene expression profiles were compared to previous single-cell gene expression experiments in human age-related macular degeneration as well as a model of laser-induced neovascularization in mice. As a secondary aim, we investigated regional gene expression patterns within the macular neural retina and between the macular and peripheral choroid.
Conclusions: Overall, this study spatially analyzes gene expression across the retina, retinal pigment epithelium, and choroid in health and describes a set of candidate molecules that become dysregulated in macular neovascularization.
Conflict of interest statement
Disclosure:
Figures



Update of
-
GENE EXPRESSION WITHIN A HUMAN CHOROIDAL NEOVASCULAR MEMBRANE USING SPATIAL TRANSCRIPTOMICS.bioRxiv [Preprint]. 2023 Jun 17:2023.06.16.544770. doi: 10.1101/2023.06.16.544770. bioRxiv. 2023. Update in: Invest Ophthalmol Vis Sci. 2023 Oct 3;64(13):40. doi: 10.1167/iovs.64.13.40. PMID: 37398429 Free PMC article. Updated. Preprint.
References
-
- Golbaz I, Ahlers C, Stock G, et al. . Quantification of the therapeutic response of intraretinal, subretinal, and subpigment epithelial compartments in exudative AMD during anti-VEGF therapy. Invest Ophthalmol Vis Sci. 2011; 52: 1599–1605. PMID 21051733. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

