Treatments During Pregnancy Targeting ERBB2 and Outcomes of Pregnant Individuals and Newborns
- PMID: 37883083
- PMCID: PMC10603505
- DOI: 10.1001/jamanetworkopen.2023.39934
Treatments During Pregnancy Targeting ERBB2 and Outcomes of Pregnant Individuals and Newborns
Abstract
Importance: Targeted therapies directed against ERBB2 are the cornerstone of medical treatment for ERBB2-positive breast cancers but are contraindicated during pregnancy.
Objectives: To describe the association of exposure to anti-ERBB2 agents during pregnancy with pregnancy and fetal or newborn outcomes, and to compare the risk and types of adverse outcomes reported more frequently in this context than after exposure to other anticancer agents.
Design, setting, and participants: For this case-control study, All reports with a pregnancy-related condition and an antineoplastic agent (Anatomical Therapeutic Chemical classification group L01) registered in the World Health Organization international pharmacovigilance database VigiBase up to June 26, 2022, were extracted. All reports with a pregnancy, an antineoplastic treatment during pregnancy, and a cancer were retained. Reports with anticancer agents prescribed for nononcologic purposes were not included.
Exposure: The exposure group was defined as reports that mention anti-ERBB2 agents compared with exposure to other anticancer agents.
Main outcome and measures: The main outcome was the reporting odds ratio (ROR) for maternofetal complications in the group exposed to anti-ERBB2 agents compared with other anticancer agents, as determined using a disproportionality analysis.
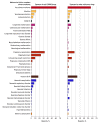
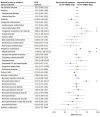
Results: A total of 3558 reports (anti-ERBB2 agents, 328; other anticancer agents, 3230) were included in the analysis. In the group exposed to anti-ERBB2 agents, most reports were from the US (159 [48.5%]), the mean (SD) age of participants was 30.8 (10.4) years, and 209 patients (97.7%) were treated for breast cancers. The molecules most frequently involved in cases with anti-ERBB2 agents were trastuzumab (n = 302), pertuzumab (n = 55), trastuzumab-emtansine (n = 20), and lapatinib (n = 18). The outcomes overreported in these cases included oligohydramnios (ROR, 17.68 [95% CI, 12.26-25.52]; P < .001), congenital respiratory tract disorders (ROR, 9.98 [95% CI, 2.88-34.67]; P < .001), and neonatal kidney failure (ROR, 9.15 [95% CI, 4.62-18.12]; P < .001). Sensitivity and multivariable analyses found similar results. Toxic effects were also significantly overreported for trastuzumab-emtansine (cardiovascular malformation: ROR, 4.46 [95% CI, 1.02-19.52]) and lapatinib (intrauterine growth restriction: ROR, 7.68 [95% CI, 3.01-19.59]).
Conclusions and relevance: In this case-control study of 328 individuals exposed to anti-ERBB2 agents during pregnancy, exposure was associated with a severe specific adverse pregnancy and fetal or newborn outcomes compared with exposure to other anticancer treatments.
Conflict of interest statement
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

