This is a preprint.
Activation of Cytotoxic Lymphocytes Through CD6 Enhances Killing of Cancer Cells
- PMID: 37886483
- PMCID: PMC10602169
- DOI: 10.21203/rs.3.rs-3405677/v1
Activation of Cytotoxic Lymphocytes Through CD6 Enhances Killing of Cancer Cells
Update in
-
Activation of cytotoxic lymphocytes through CD6 enhances killing of cancer cells.Cancer Immunol Immunother. 2024 Jan 27;73(2):34. doi: 10.1007/s00262-023-03578-1. Cancer Immunol Immunother. 2024. PMID: 38280067 Free PMC article.
Abstract
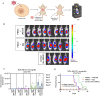
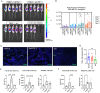
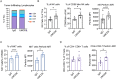
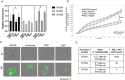
Immune checkpoint inhibitors (ICIs) have demonstrated efficacy and improved survival in a growing number of cancers. Despite their success, ICIs are associated with immune-related adverse events that can interfere with their use. Therefore, safer approaches are needed. CD6, expressed by T-lymphocytes and human NK cells, engages in cell-cell interactions by binding to its ligands CD166 (ALCAM) and CD318 (CDCP1). CD6 is a target protein for regulating immune responses and is required for the development of several mouse models of autoimmunity. Interestingly, CD6 is exclusively expressed on immune cells while CD318 is strongly expressed on most cancers. Here we demonstrate that disrupting the CD6-CD318 axis with UMCD6, an anti-CD6 monoclonal antibody, prolongs survival of mice in xenograft models of human breast and prostate cancer, treated with infusions of human lymphocytes. Analysis of tumor-infiltrating immune cells showed that augmentation of lymphocyte cytotoxicity by UMCD6 is due to effects of this antibody on NK, NKT and CD8+ T cells. Tumor-infiltrating cytotoxic lymphocytes were found in higher proportions and were activated in UMCD6-treated mice compared to controls. Similar changes in gene expression were observed by RNA-seq analysis of NK cells treated with UMCD6. Particularly, UMCD6 up-regulated the NKG2D-DAP10 complex and activated PI3K. Thus, the CD6-CD318 axis can regulate the activation state of cytotoxic lymphocytes and their positioning within the tumor microenvironment.
Keywords: CD6; NK; cytotoxic lymphocyte; immunotherapy.
Conflict of interest statement
Competing Interests: Drs. Fox and Lin hold equity in Abcon, a company created to test the use of therapeutic agents that interrupt the interactions between CD6 and CD6 ligands.
Figures


References
-
- Martinez VG, Moestrup SK, Holmskov U, Mollenhauer J, Lozano F. The conserved scavenger receptor cysteine-rich superfamily in therapy and diagnosis. Pharmacol Rev. 2011; 63(4):967–1000. - PubMed
-
- Braun M, Muller B, ter Meer D, Raffegerst S, Simm B, Wilde S, Spranger S, Ellwart J, Mosetter B, Umansky L, Lerchl T, Schendel DJ, Falk CS. The CD6 scavenger receptor is differentially expressed on a CD56 natural killer cell subpopulation and contributes to natural killer-derived cytokine and chemokine secretion. J Innate Immun. 2011; 3(4):420–434. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

