Monoclonal Antibody Disrupts Biofilm Structure and Restores Antibiotic Susceptibility in an Orthopedic Implant Infection Model
- PMID: 37887191
- PMCID: PMC10604051
- DOI: 10.3390/antibiotics12101490
Monoclonal Antibody Disrupts Biofilm Structure and Restores Antibiotic Susceptibility in an Orthopedic Implant Infection Model
Abstract
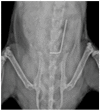
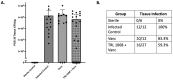
Bacterial biofilms on orthopedic implants are resistant to the host immune response and to traditional systemic antibiotics. Novel therapies are needed to improve patient outcomes. TRL1068 is a human monoclonal antibody (mAb) against a biofilm anchoring protein. For assessment of this agent in an orthopedic implant infection model, efficacy was measured by reduction in bacterial burden of Staphylococcus aureus, the most common pathogen for prosthetic joint infections (PJI). Systemic treatment with the biofilm disrupting mAb TRL1068 in conjunction with vancomycin eradicated S. aureus from steel pins implanted in the spine for 26 of 27 mice, significantly more than for vancomycin alone. The mechanism of action was elucidated by two microscopy studies. First, TRL1068 was localized to biofilm using a fluorescent antibody tag. Second, a qualitative effect on biofilm structure was observed using scanning electron microscopy (SEM) to examine steel pins that had been treated in vivo. SEM images of implants retrieved from control mice showed abundant three-dimensional biofilms, whereas those from mice treated with TRL1068 did not. Clinical Significance: TRL1068 binds at high affinity to S. aureus biofilms, thereby disrupting the three-dimensional structure and significantly reducing implant CFUs in a well-characterized orthopedic model for which prior tested agents have shown only partial efficacy. TRL1068 represents a promising systemic treatment for orthopedic implant infection.
Keywords: S. aureus; biofilm; monoclonal antibody; orthopedic implant infection; prosthetic joint infection; spinal implant infection.
Conflict of interest statement
N.M.B. reports consulting fees from Biomet, Bone Support, Daiichi Sankyo, and Onkos, and he is a board or committee member of the Musculoskeletal Tumor Society and Orthopaedic Research and Education Foundation. L.M.K. and E.T. are paid employees or consultants of Trellis Bioscience, Inc. All other authors report no relevant competing interests.
Figures



References
-
- Saeed K., McLaren A.C., Schwarz E.M., Antoci V., Arnold W.V., Chen A.F., Clauss M., Esteban J., Gant V., Hendershot E., et al. 2018 international consensus meeting on musculoskeletal infection: Summary from the biofilm workgroup and consensus on biofilm related musculoskeletal infections. J. Orthop. Res. 2019;37:1007–1017. doi: 10.1002/jor.24229. - DOI - PubMed

