Ssn6 Interacts with Polar Tube Protein 2 and Transcriptional Repressor for RNA Polymerase II: Insight into Its Involvement in the Biological Process of Microsporidium Nosema bombycis
- PMID: 37888246
- PMCID: PMC10608102
- DOI: 10.3390/jof9100990
Ssn6 Interacts with Polar Tube Protein 2 and Transcriptional Repressor for RNA Polymerase II: Insight into Its Involvement in the Biological Process of Microsporidium Nosema bombycis
Abstract
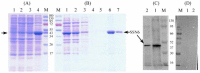
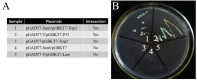
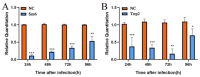
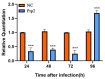
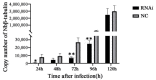
Nosema bombycis is a representative species of Microsporidia, and is the pathogen that causes pebrine disease in silkworms. In the process of infection, the polar tube of N. bombycis is injected into the host cells. During proliferation, N. bombycis recruits the mitochondria of host cells. The general transcriptional corepressor Ssn6 contains six tetratricopeptide repeats (TPR) and undertakes various important functions. In this study, we isolated and characterized Nbssn6 of the microsporidium N. bombycis. The Nbssn6 gene contains a complete ORF of 1182 bp in length that encodes a 393 amino acid polypeptide. Indirect immunofluorescence assay showed that the Ssn6 protein was mainly distributed in the cytoplasm and nucleus at the proliferative phase of N. bombycis. We revealed the interaction of Nbssn6 with polar tube protein 2 (Nbptp2) and the transcriptional repressor for RNA polymerase II (Nbtrrp2) by Co-IP and yeast two-hybrid assays. Results from RNA interference further confirmed that the transcriptional level of Nbptp2 and Nbtrrp2 was regulated by Nbssn6. These results suggest that Nbssn6 impacts the infection and proliferation of N. bombycis via interacting with the polar tube protein and transcriptional repressor for RNA polymerase II.
Keywords: general transcriptional corepressor; microsporidia; polar tube protein; transcriptional repressor for RNA polymerase II.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







Similar articles
-
Identification, expression and subcellular localization of Orc1 in the microsporidian Nosema bombycis.Gene. 2022 Aug 5;834:146607. doi: 10.1016/j.gene.2022.146607. Epub 2022 May 21. Gene. 2022. PMID: 35609797
-
Proteomic profile of polar filament and polar tube from fungal pathogen microsporidium Nosema bombycis provides new insights into its unique invasion organelle.J Proteomics. 2022 Jul 15;263:104617. doi: 10.1016/j.jprot.2022.104617. Epub 2022 May 18. J Proteomics. 2022. PMID: 35595055
-
Sar1 Interacts with Sec23/Sec24 and Sec13/Sec31 Complexes: Insight into Its Involvement in the Assembly of Coat Protein Complex II in the Microsporidian Nosema bombycis.Microbiol Spectr. 2022 Dec 21;10(6):e0071922. doi: 10.1128/spectrum.00719-22. Epub 2022 Oct 27. Microbiol Spectr. 2022. PMID: 36301095 Free PMC article.
-
Nosema bombycis: A remarkable unicellular parasite infecting insects.J Eukaryot Microbiol. 2024 Sep-Oct;71(5):e13045. doi: 10.1111/jeu.13045. Epub 2024 Aug 2. J Eukaryot Microbiol. 2024. PMID: 39095558 Review.
-
The roles of microsporidia spore wall proteins in the spore wall formation and polar tube anchorage to spore wall during development and infection processes.Exp Parasitol. 2018 Apr;187:93-100. doi: 10.1016/j.exppara.2018.03.007. Epub 2018 Mar 6. Exp Parasitol. 2018. PMID: 29522765 Review.
Cited by
-
The Function of Different Subunits of the Molecular Chaperone CCT in the Microsporidium Nosema bombycis: NbCCTζ Interacts with NbCCTα.J Fungi (Basel). 2024 Mar 20;10(3):229. doi: 10.3390/jof10030229. J Fungi (Basel). 2024. PMID: 38535237 Free PMC article.
References
-
- Higes M., Martin-Hernandez R., Botias C., Bailon E.G., Gonzalez-Porto A.V., Barrios L., Del Nozal M.J., Bernal J.L., Jimenez J.J., Palencia P.G., et al. How natural infection by Nosema ceranae causes honeybee colony collapse. Environ. Microbiol. 2008;10:2659–2669. doi: 10.1111/j.1462-2920.2008.01687.x. - DOI - PubMed
LinkOut - more resources
Full Text Sources

