The Role of Cyclin-Dependent Kinases (CDK) 4/6 in the Ovarian Tissue and the Possible Effects of Their Exogenous Inhibition
- PMID: 37894292
- PMCID: PMC10605229
- DOI: 10.3390/cancers15204923
The Role of Cyclin-Dependent Kinases (CDK) 4/6 in the Ovarian Tissue and the Possible Effects of Their Exogenous Inhibition
Abstract
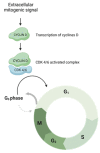
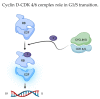
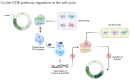
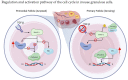
The combination of cyclin-dependent kinase (CDK) 4/6 inhibitors with endocrine therapy is the standard treatment for patients with HR+/HER2- advanced breast cancer. Recently, this combination has also entered the early setting as an adjuvant treatment in patients with HR+/HER2- disease at a high risk of disease recurrence following (neo)adjuvant chemotherapy. Despite their current use in clinical practice, limited data on the potential gonadotoxicity of CDK4/6 inhibitors are available. Hence, fully informed treatment decision making by premenopausal patients concerned about the potential development of premature ovarian insufficiency and infertility with the proposed therapy remains difficult. The cell cycle progression of granulosa and cumulus cells is a critical process for ovarian function, especially for ensuring proper follicular growth and acquiring competence. Due to the pharmacological properties of CDK4/6 inhibitors, there could be a potentially negative impact on ovarian function and fertility in women of reproductive age. This review aims to summarize the role of the cyclin D-CDK4 and CDK6 complexes in the ovary and the potential impact of CDK4/6 inhibition on its physiological processes.
Keywords: CDK 4/6 inhibitors; cyclin D-CDK 4/6 complex; gonadotoxicity; ovary.
Conflict of interest statement
Matteo Lambertini reports his advisory role for Roche, Lilly, Novartis, Astrazeneca, Pfizer, Seagen, Gilead, MSD and Exact Sciences; speaker honoraria from Roche, Lilly, Novartis, Pfizer, Sandoz, Libbs, Daiichi Sankyo and Takeda; travel grants from Gilead and Daiichi Sankyo; and research support (to the Institution) from Gilead outside the submitted work. Eva Blondeaux reports research support (to the Institution) from Gilead outside the submitted work. All the other authors declare no conflicts of interest.
Figures
References
-
- Paluch-Shimon S., Cardoso F., Partridge A.H., Abulkhair O., Azim H.A., Bianchi-Micheli G., Cardoso M.J., Curigliano G., Gelmon K.A., Gentilini O., et al. ESO-ESMO Fifth International Consensus Guidelines for Breast Cancer in Young Women (BCY5) Ann. Oncol. 2022;33:1097–1118. doi: 10.1016/j.annonc.2022.07.007. - DOI - PubMed
-
- Lambertini M., Peccatori F.A., Demeestere I., Amant F., Wyns C., Stukenborg J.-B., Paluch-Shimon S., Halaska M.J., Uzan C., Meissner J., et al. Fertility Preservation and Post-Treatment Pregnancies in Post-Pubertal Cancer Patients: ESMO Clinical Practice Guidelines. Ann. Oncol. 2020;31:1664–1678. doi: 10.1016/j.annonc.2020.09.006. - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

