A Pharmacological Investigation of Eph-Ephrin Antagonism in Prostate Cancer: UniPR1331 Efficacy Evidence
- PMID: 37895923
- PMCID: PMC10609876
- DOI: 10.3390/ph16101452
A Pharmacological Investigation of Eph-Ephrin Antagonism in Prostate Cancer: UniPR1331 Efficacy Evidence
Abstract
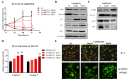
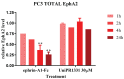
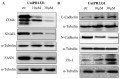
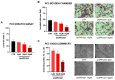
The Eph kinases are the largest receptor tyrosine kinases (RTKs) family in humans. PC3 human prostate adenocarcinoma cells are a well-established model for studying Eph-ephrin pharmacology as they naturally express a high level of EphA2, a promising target for new cancer therapies. A pharmacological approach with agonists did not show significant efficacy on tumor growth in prostate orthotopic murine models, but reduced distal metastasis formation. In order to improve the comprehension of the pharmacological targeting of Eph receptors in prostate cancer, in the present work, we investigated the efficacy of Eph antagonism both in vitro and in vivo, using UniPR1331, a small orally bioavailable Eph-ephrin interaction inhibitor. UniPR1331 was able to inhibit PC3 cells' growth in vitro in a dose-dependent manner, affecting the cell cycle and inducing apoptosis. Moreover, UniPR1331 promoted the PC3 epithelial phenotype, downregulating epithelial mesenchymal transition (EMT) markers. As a consequence, UniPR1331 reduced in vitro PC3 migration, invasion, and vasculomimicry capabilities. The antitumor activity of UniPR1331 was confirmed in vivo when administered alone or in combination with cytotoxic drugs in PC3-xenograft mice. Our results demonstrated that Eph antagonism is a promising strategy for inhibiting prostate cancer growth, especially in combination with cytotoxic drugs.
Keywords: Eph receptors; EphA2 antagonism; PC3 xenograft mice; ephrin ligands; prostate cancer.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
Similar articles
-
UniPR1331: Small Eph/Ephrin Antagonist Beneficial in Intestinal Inflammation by Interfering with Type-B Signaling.Pharmaceuticals (Basel). 2021 May 24;14(6):502. doi: 10.3390/ph14060502. Pharmaceuticals (Basel). 2021. PMID: 34074058 Free PMC article.
-
UniPR1331, a small molecule targeting Eph/ephrin interaction, prolongs survival in glioblastoma and potentiates the effect of antiangiogenic therapy in mice.Oncotarget. 2018 May 11;9(36):24347-24363. doi: 10.18632/oncotarget.25272. eCollection 2018 May 11. Oncotarget. 2018. PMID: 29849945 Free PMC article.
-
Lithocholic acid is an Eph-ephrin ligand interfering with Eph-kinase activation.PLoS One. 2011 Mar 30;6(3):e18128. doi: 10.1371/journal.pone.0018128. PLoS One. 2011. PMID: 21479221 Free PMC article.
-
May EPH/Ephrin Targeting Revolutionize Lung Cancer Treatment?Int J Mol Sci. 2022 Dec 21;24(1):93. doi: 10.3390/ijms24010093. Int J Mol Sci. 2022. PMID: 36613532 Free PMC article. Review.
-
Protein-Protein Interaction Inhibitors Targeting the Eph-Ephrin System with a Focus on Amino Acid Conjugates of Bile Acids.Pharmaceuticals (Basel). 2022 Jan 24;15(2):137. doi: 10.3390/ph15020137. Pharmaceuticals (Basel). 2022. PMID: 35215250 Free PMC article. Review.
References
-
- Kosinski C., Li V.S.W., Chan A.S.Y., Zhang J., Ho C., Tsui W.Y., Chan T.L., Mifflin R.C., Powell D.W., Yuen S.T., et al. Gene Expression Patterns of Human Colon Tops and Basal Crypts and BMP Antagonists as Intestinal Stem Cell Niche Factors. Proc. Natl. Acad. Sci. USA. 2007;104:15418–15423. doi: 10.1073/pnas.0707210104. - DOI - PMC - PubMed
-
- Ji H., Goode R.J.A., Vaillant F., Mathivanan S., Kapp E.A., Mathias R.A., Lindeman G.J., Visvader J.E., Simpson R.J. Proteomic Profiling of Secretome and Adherent Plasma Membranes from Distinct Mammary Epithelial Cell Subpopulations. Proteomics. 2011;11:4029–4039. doi: 10.1002/pmic.201100102. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

