Elevated CO2 Can Worsen Fusarium Head Blight Disease Severity in Wheat but the Fhb1 QTL Provides Reliable Disease Resistance
- PMID: 37895995
- PMCID: PMC10610529
- DOI: 10.3390/plants12203527
Elevated CO2 Can Worsen Fusarium Head Blight Disease Severity in Wheat but the Fhb1 QTL Provides Reliable Disease Resistance
Abstract
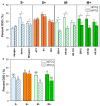
Fusarium head blight (FHB) is a destructive fungal disease of wheat that causes significant economic loss due to lower yields and the contamination of grain with fungal toxins (mycotoxins), particularly deoxynivalenol (DON). FHB disease spread and mycotoxin contamination has been shown to worsen at elevated CO2, therefore, it is important to identify climate-resilient FHB resistance. This work evaluates whether wheat with the Fhb1 quantitative trait locus (QTL), the most widely deployed FHB resistance locus in wheat breeding programs, provides reliable disease resistance at elevated CO2. Near-isogenic wheat lines (NILs) derived from either a highly FHB susceptible or a more FHB resistant genetic background, with or without the Fhb1 QTL, were grown in growth chambers at ambient (400 ppm) and elevated (1000 ppm) CO2 conditions. Wheat was inoculated with Fusarium graminearum and evaluated for FHB severity. At elevated CO2, the NILs derived from more FHB-resistant wheat had increased disease spread, greater pathogen biomass and mycotoxin contamination, and lower rates of DON detoxification; this was not observed in wheat from a FHB susceptible genetic background. The Fhb1 QTL was not associated with increased disease severity in wheat grown at elevated CO2 and provided reliable disease resistance.
Keywords: Fhb1; climate resilience; elevated CO2; food safety; fusarium head blight; mycotoxins; wheat.
Conflict of interest statement
Authors have no conflict of interest to declare. USDA is an equal opportunity provider and employer. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Figures




References
-
- O’Donnell K., Ward T.J., Geiser D.M., Kistler H.C., Aoki T. Genealogical concordance between the mating type locus and seven other nuclear genes supports formal recognition of nine phylogenetically distinct species within the Fusarium graminearum clade. Fungal Genet. Biol. 2004;41:600–623. doi: 10.1016/j.fgb.2004.03.003. - DOI - PubMed
-
- Ward T.J., Clear R.M., Rooney A.P., O’Donnell K., Gaba D., Patrick S., Starkey D.E., Gilbert J., Geiser D.M., Nowicki T.W. An adaptive evolutionary shift in Fusarium head blight pathogen populations is driving the rapid spread of more toxigenic Fusarium graminearum in North America. Fungal Genet. Biol. 2008;45:473–484. doi: 10.1016/j.fgb.2007.10.003. - DOI - PubMed
LinkOut - more resources
Full Text Sources

