Systemic Dendrimer-Peptide Therapies for Wet Age-Related Macular Degeneration
- PMID: 37896188
- PMCID: PMC10609940
- DOI: 10.3390/pharmaceutics15102428
Systemic Dendrimer-Peptide Therapies for Wet Age-Related Macular Degeneration
Abstract
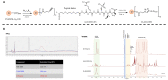
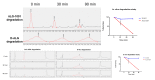
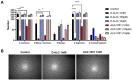
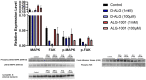
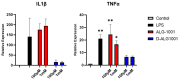
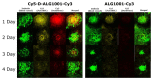
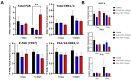
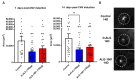
Wet age-related macular degeneration (AMD) is an end-stage event in a complex pathogenesis of macular degeneration, involving the abnormal growth of blood vessels at the retinal pigment epithelium driven by vascular endothelial growth factor (VEGF). Current therapies seek to interrupt VEGF signaling to halt the progress of neovascularization, but a significant patient population is not responsive. New treatment modalities such as integrin-binding peptides (risuteganib/Luminate/ALG-1001) are being explored to address this clinical need but these treatments necessitate the use of intravitreal injections (IVT), which carries risks of complications and restricts its availability in less-developed countries. Successful systemic delivery of peptide-based therapeutics must overcome obstacles such as degradation by proteinases in circulation and off-target binding. In this work, we present a novel dendrimer-integrin-binding peptide (D-ALG) synthesized with a noncleavable, "clickable" linker. In vitro, D-ALG protected the peptide payload from enzymatic degradation for up to 1.5 h (~90% of the compound remained intact) in a high concentration of proteinase (2 mg/mL) whereas ~90% of free ALG-1001 was degraded in the same period. Further, dendrimer conjugation preserved the antiangiogenic activity of ALG-1001 in vitro with significant reductions in endothelial vessel network formation compared to untreated controls. In vivo, direct intravitreal injections of ALG-1001 and D-ALG produced reductions in the CNV lesion area but in systemically dosed animals, only D-ALG produced significant reductions of CNV lesion area at 14 days. Imaging data suggested that the difference in efficacy may be due to more D-ALG remaining in the target area than ALG-1001 after administration. The results presented here offer a clinically relevant route for peptide therapeutics by addressing the major obstacles that these therapies face in delivery.
Keywords: AMD; hydroxyl PAMAM dendrimer; integrin; neovascularization; peptide conjugates; targeted delivery.
Conflict of interest statement
T.W and R.M.K. are co-inventors are patents relating to the compositions described in this manuscript.
Figures
Similar articles
-
Systemic dendrimer nanotherapies for targeted suppression of choroidal inflammation and neovascularization in age-related macular degeneration.J Control Release. 2021 Jul 10;335:527-540. doi: 10.1016/j.jconrel.2021.05.035. Epub 2021 May 29. J Control Release. 2021. PMID: 34058271
-
Risuteganib-a novel integrin inhibitor for the treatment of non-exudative (dry) age-related macular degeneration and diabetic macular edema.Expert Opin Investig Drugs. 2020 Jun;29(6):547-554. doi: 10.1080/13543784.2020.1763953. Epub 2020 May 13. Expert Opin Investig Drugs. 2020. PMID: 32349559 Review.
-
Differential effects of risuteganib and bevacizumab on AMD cybrid cells.Exp Eye Res. 2021 Feb;203:108287. doi: 10.1016/j.exer.2020.108287. Epub 2020 Oct 16. Exp Eye Res. 2021. PMID: 33075294 Free PMC article.
-
An efficacy analysis of anti-vascular endothelial growth factor therapy for choroidal neovascularization secondary to multifocal choroiditis and comparison with wet age-related macular degeneration.J Zhejiang Univ Sci B. 2018 Apr.;19(4):327-332. doi: 10.1631/jzus.B1700535. J Zhejiang Univ Sci B. 2018. PMID: 29616508 Free PMC article.
-
Clinical evidence of intravitreal triamcinolone acetonide in the management of age-related macular degeneration.Curr Drug Targets. 2011 Feb;12(2):149-72. doi: 10.2174/138945011794182746. Curr Drug Targets. 2011. PMID: 20887246 Review.
Cited by
-
Ocular drug delivery systems based on nanotechnology: a comprehensive review for the treatment of eye diseases.Discov Nano. 2025 May 3;20(1):75. doi: 10.1186/s11671-025-04234-6. Discov Nano. 2025. PMID: 40317427 Free PMC article. Review.
-
Dendrimers: A Themed Issue in Honor of Professor Donald A. Tomalia on the Occasion of His 85th Birthday, Recognizing His Outstanding Achievements in Advancing the Field of Dendrimers.Pharmaceutics. 2024 Dec 17;16(12):1608. doi: 10.3390/pharmaceutics16121608. Pharmaceutics. 2024. PMID: 39771586 Free PMC article.
-
OcuPair, a Novel Photo-crosslinkable PAMAM Dendrimer-Hyaluronic Acid Hydrogel Bandage/Bioadhesive for Corneal Injuries and Temporary Corneal Repair.Adv Sci (Weinh). 2025 Jun;12(23):e2417731. doi: 10.1002/advs.202417731. Epub 2025 Mar 27. Adv Sci (Weinh). 2025. PMID: 40145837 Free PMC article.
-
Utilization of Nanoparticles for Treating Age-Related Macular Degeneration.Pharmaceuticals (Basel). 2025 Jan 25;18(2):162. doi: 10.3390/ph18020162. Pharmaceuticals (Basel). 2025. PMID: 40005976 Free PMC article. Review.
-
Dendrimers, Dendrons, and the Dendritic State: Reflection on the Last Decade with Expected New Roles in Pharma, Medicine, and the Life Sciences.Pharmaceutics. 2024 Nov 28;16(12):1530. doi: 10.3390/pharmaceutics16121530. Pharmaceutics. 2024. PMID: 39771509 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

