The Path from Nasal Tissue to Nasal Mucosa on Chip: Part 2-Advanced Microfluidic Nasal In Vitro Model for Drug Absorption Testing
- PMID: 37896199
- PMCID: PMC10610000
- DOI: 10.3390/pharmaceutics15102439
The Path from Nasal Tissue to Nasal Mucosa on Chip: Part 2-Advanced Microfluidic Nasal In Vitro Model for Drug Absorption Testing
Abstract
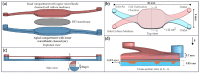
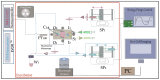
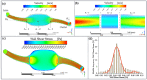
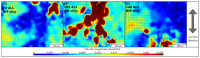
The nasal mucosa, being accessible and highly vascularized, opens up new opportunities for the systemic administration of drugs. However, there are several protective functions like the mucociliary clearance, a physiological barrier which represents is a difficult obstacle for drug candidates to overcome. For this reason, effective testing procedures are required in the preclinical phase of pharmaceutical development. Based on a recently reported immortalized porcine nasal epithelial cell line, we developed a test platform based on a tissue-compatible microfluidic chip. In this study, a biomimetic glass chip, which was equipped with a controlled bidirectional airflow to induce a physiologically relevant wall shear stress on the epithelial cell layer, was microfabricated. By developing a membrane transfer technique, the epithelial cell layer could be pre-cultivated in a static holder prior to cultivation in a microfluidic environment. The dynamic cultivation within the chip showed a homogenous distribution of the mucus film on top of the cell layer and a significant increase in cilia formation compared to the static cultivation condition. In addition, the recording of the ciliary transport mechanism by microparticle image velocimetry was successful. Using FITC-dextran 4000 as an example, it was shown that this nasal mucosa on a chip is suitable for permeation studies. The obtained permeation coefficient was in the range of values determined by means of other established in vitro and in vivo models. This novel nasal mucosa on chip could, in future, be automated and used as a substitute for animal testing.
Keywords: microfluidics; mucociliary clearance; nasal drug administration; nasal mucosa; organ on chip; permeation test.
Conflict of interest statement
The Authors Tobias May and Kristina Nehlsen are/were employed by InSCREENeX GmbH, Germany. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflicts of interest.
Figures




Similar articles
-
The Path from Nasal Tissue to Nasal Mucosa on Chip: Part 1-Establishing a Nasal In Vitro Model for Drug Delivery Testing Based on a Novel Cell Line.Pharmaceutics. 2023 Aug 30;15(9):2245. doi: 10.3390/pharmaceutics15092245. Pharmaceutics. 2023. PMID: 37765214 Free PMC article.
-
In vitro interactions of aerosol formulations with human nasal epithelium using real-time monitoring of drug transport in a nasal mucosa-on-a-chip.Biosens Bioelectron. 2023 Mar 1;223:115010. doi: 10.1016/j.bios.2022.115010. Epub 2022 Dec 17. Biosens Bioelectron. 2023. PMID: 36586150
-
Real-time quantitative monitoring of in vitro nasal drug delivery by a nasal epithelial mucosa-on-a-chip model.Expert Opin Drug Deliv. 2021 Jun;18(6):803-818. doi: 10.1080/17425247.2021.1873274. Epub 2021 Jan 19. Expert Opin Drug Deliv. 2021. PMID: 33410717
-
The effect of cilia and the mucociliary clearance on successful drug delivery.Biol Pharm Bull. 2015;38(4):497-506. doi: 10.1248/bpb.b14-00398. Epub 2015 Feb 7. Biol Pharm Bull. 2015. PMID: 25739664 Review.
-
Intestinal Models for Personalized Medicine: from Conventional Models to Microfluidic Primary Intestine-on-a-chip.Stem Cell Rev Rep. 2022 Aug;18(6):2137-2151. doi: 10.1007/s12015-021-10205-y. Epub 2021 Jun 28. Stem Cell Rev Rep. 2022. PMID: 34181185 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

