New-Onset Rheumatic Immune-Mediated Inflammatory Diseases Following SARS-CoV-2 Vaccinations until May 2023: A Systematic Review
- PMID: 37896974
- PMCID: PMC10610967
- DOI: 10.3390/vaccines11101571
New-Onset Rheumatic Immune-Mediated Inflammatory Diseases Following SARS-CoV-2 Vaccinations until May 2023: A Systematic Review
Abstract
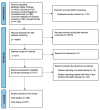
A comprehensive, up-to-date systematic review (SR) of the new-onset rheumatic immune-mediated inflammatory diseases (R-IMIDs) following COVID-19 vaccinations is lacking. Therefore, we investigated the demographics, management, and prognosis of new R-IMIDs in adults following SARS-CoV-2 vaccinations. A systematic literature search of Medline, Embase, Google Scholar, LitCovid, and Cochrane was conducted. We included any English-language study that reported new-onset R-IMID in adults following the post-COVID-19 vaccination. A total of 271 cases were reported from 39 countries between January 2021 and May 2023. The mean age of patients was 56 (range 18-90), and most were females (170, 62.5%). Most (153, 56.5%) received the Pfizer BioNTech COVID-19 vaccine. Nearly 50% of patients developed R-IMID after the second dose of the vaccine. Vasculitis was the most prevalent clinical presentation (86, 31.7%), followed by connective tissue disease (66, 24.3%). The mean duration between the vaccine's 'trigger' dose and R-IMID was 11 days. Most (220, 81.2%) received corticosteroids; however, 42% (115) received DMARDs such as methotrexate, cyclophosphamide, tocilizumab, anakinra, IV immunoglobulins, plasma exchange, or rituximab. Complete remission was achieved in 75 patients (27.7%), and 137 (50.6%) improved following the treatment. Two patients died due to myositis. This SR highlights that SARS-CoV-2 vaccines may trigger R-IMID; however, further epidemiology studies are required.
Keywords: COVID-19 vaccines; adverse events following immunization; connective tissue diseases; immune-mediated inflammatory disease; inflammatory arthritis; rheumatic disease; vasculitis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Cascella M., Rajnik M., Aleem A., Dulebohn S.C., Napoli R.D. Features, Evaluation, and Treatment of Coronavirus (COVID-19) StatPearls—NCBI Bookshelf; Treasure Island, FL, USA: 2023. [(accessed on 15 September 2023)]. Available online: https://www.ncbi.nlm.nih.gov/books/NBK554776/ - PubMed
-
- Drugs and Lactation Database (LactMed®®) COVID-19 Vaccines—NCBI Bookshelf; Treasure Island, FL, USA: 2023. [(accessed on 15 September 2023)]. Available online: https://www.ncbi.nlm.nih.gov/books/NBK565969/
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous