Patient-reported Outcomes and Quality of Life in Anemic and Symptomatic Patients With Myelofibrosis: Results From the MOMENTUM Study
- PMID: 37901848
- PMCID: PMC10599984
- DOI: 10.1097/HS9.0000000000000966
Patient-reported Outcomes and Quality of Life in Anemic and Symptomatic Patients With Myelofibrosis: Results From the MOMENTUM Study
Abstract
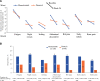
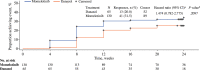
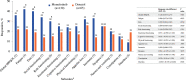
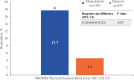
Myelofibrosis (MF) is a chronic myeloproliferative neoplasm that typically manifests with debilitating symptoms that progressively worsen, negatively impacting patients' quality of life. Fatigue is a multifactorial and burdensome MF-related symptom due to its severity, persistence, and prevalence, with anemia a contributing factor and major unmet need. Clinical trials of the Janus kinase (JAK)1/JAK2/activin A receptor type 1 inhibitor momelotinib have shown consistent anemia benefits, in addition to improvements in MF-related symptoms. The phase 3 MOMENTUM trial in symptomatic and anemic patients met its primary end point, with a greater proportion having a Myelofibrosis Symptom Assessment Form (MFSAF) Total Symptom Score (TSS) reduction ≥50% at week 24 with momelotinib versus danazol. To support the positive primary end point result, we conducted longitudinal, responder, and time-to-event analyses of patient-reported outcomes from MOMENTUM, as measured by the MFSAF, European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30), and Patient-Reported Outcomes Measurement Information System (PROMIS) assessments. These analyses demonstrated rapid and durable response benefits with momelotinib, with achievement of first TSS response by day 29 and continued improvement over time. Improvements favored momelotinib versus danazol for each MFSAF individual item, and greater improvements were observed for disease- and cancer-related fatigue and physical functioning at week 24, with significant results for multiple items/domains across the 3 assessments. These findings are consistent in demonstrating that momelotinib provides substantial symptom benefit.
Copyright © 2023 the Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the European Hematology Association.
Conflict of interest statement
RAM has consulted and received honoraria from Novartis, Sierra Oncology, Gentech, Blueprint, Geron, Telios, CTI, Incyte, BMS, AbbVie, GSK and Morphosys. CH has consulted for Galecto DISC and Keros and received compensation; received honoraria from Novartis, CTI, BMS, Sierra, and GSK; participated on an Ad Board for AOP, Telios, Sumitomo, and Keros; and had a leadership role in EHA, HemaSphere, and MPN Voice. VG has consulted for Novartis, BMS Celgene, Sierra Oncology, AbbVie, Constellation Biopharma, Pfizer, GSK Pharma, and CTI Biopharma and received compensation; received honoraria from Novartis and BMS Celgene; and received compensation for participation on an Ad Board for BMS Celgene, Roche, AbbVie, Pfizer, Sierra Oncology, and CTI Biopharma. DPM received a research grant from Imago Biosciences; received honoraria from JAZZ Pharma, AbbVie, and Novartis; participated in the UK ALL RIC TRIAL – DSM board and is an EBMT Scientific Council Member Chair of the EBMT CMWP. MFM received honoraria from Novartis, Abbvie, and AOP; and received compensation for participation on an Ad Board from Novartis, BMS, AbbVie, CTI, Sierra Oncology, and GSK. J-JK received compensation and participated in Ad Boards for Novartis, AbbVie, BMS, GSK, Incyte, and AOP Health. LF received honoraria from Novartis and BMS and participated in Ad Boards for Incyte and GS. MLF has consulted for Novartis, GSK, AbbVie, and Sierra Oncology and received compensation; and received honoraria from Novartis and BMS and attended meetings for AbbVie. DMR has consulted for Keros, and the institution received compensation; received honoraria from Novartis personally and to the institution; received compensation from Novartis to attend a meeting; received compensation to the institution for participation on an Ad Board from Novartis, Menarini, and Takeda. STO has consulted for AbbVie, Constellation, CTI BioPharma, BMS, Geron, Sierra Oncology, Cogent, Protagonist, and Incyte and received compensation. JK owned stock in Sierra Oncology and owns stock in Gilead. SR and RD owned stock in Sierra Oncology. SD is an employee at Sierra Oncology. BG received compensation from GSK for attending meetings and owns stock in GSK. SV received compensation for participation on an Ad Board. UP received honoraria and research support from Geron, GSK, Novartis, Abbvie, and Curis. JMP received honoraria to the institution from CTI; and received compensation to the institution for participation in an Ad Board for Morphosys. AJM received support from Abbvie for the present article; received grants or contracts from Celgene/BMS; received royalties or licenses from Alethiomics; received consulting fees from Celgene/BMS paid to the Sierra Oncology, Novartis paid to Karyopharm, Abbvie paid to Sensyn, and CTI paid to Incyte, Galecto, Pfizer, and Gilead; received honoraria from Celgene/BMS, Novartis, and Abbvie; received support to attend meetings from Celgene/BMS and Novartis; and participated on an Ad Board for Celgene/BMS and Abbvie. All authors received medical writing support for this article funded by Sierra Oncology, a GSK company. All the other authors have no conflicts of interest to disclose.
Figures



References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
