Adopting human factors in early phase and experimental medicine research: A nested pilot study observing controlled human infection with SARS-CoV-2
- PMID: 37903635
- PMCID: PMC11497241
- DOI: 10.1111/bcp.15949
Adopting human factors in early phase and experimental medicine research: A nested pilot study observing controlled human infection with SARS-CoV-2
Abstract
Aims: The influence of human factors on safety in healthcare settings is well established, with targeted interventions reducing risk and enhancing team performance. In experimental and early phase clinical research participant safety is paramount and safeguarded by guidelines, protocolized care and staff training; however, the real-world interaction and implementation of these risk-mitigating measures has never been subjected to formal system-based assessment.
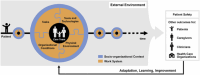
Methods: Independent structured observations, systematic review of study documents, and interviews and focus groups were used to collate data on three key tasks undertaken in a clinical research facility (CRF) during a SARS CoV-2 controlled human infection model (CHIM) study. The Systems Engineering Initiative for Patient Safety (SEIPS) was employed to analyse and categorize findings, and develop recommendations for safety interventions.
Results: High levels of team functioning and a clear focus on participant safety were evident throughout the study. Despite this, latent risks in both study-specific and CRF work systems were identified in all four SEIPS domains (people, environment, tasks and tools). Fourteen actionable recommendations were generated collaboratively. These included inter-organization and inter-study standardization, optimized checklists for safety critical tasks, and use of simulation for team training and exploration of work systems.
Conclusions: This pioneering application of human factors techniques to analyse work systems during the conduct of research in a CRF revealed risks unidentified by routine review and appraisal, and despite international guideline adherence. SEIPS may aid categorization of system problems and the formulation of recommendations that reduce risk and mitigate potential harm applicable across a trials portfolio.
Keywords: clinical trials; human factors; methodology; patient safety; translational research.
© 2023 The Authors. British Journal of Clinical Pharmacology published by John Wiley & Sons Ltd on behalf of British Pharmacological Society.
Conflict of interest statement
R.L.R. and S.J. have previously contributed to intellectual property licensed by Oxford University Innovation to AstraZeneca. All authors declared no competing interests for this work.
Figures
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

