Define of Optimal Addition Period of Osteogenic Peptide to Accelerate the Osteogenic Differentiation of Human Pluripotent Stem Cells
- PMID: 37903982
- PMCID: PMC10825087
- DOI: 10.1007/s13770-023-00597-y
Define of Optimal Addition Period of Osteogenic Peptide to Accelerate the Osteogenic Differentiation of Human Pluripotent Stem Cells
Abstract
Background: The addition of growth factiors is commonly applied to improve the osteogenic differentiation of stem cells. However, for human pluripotent stem cells (hPSCs), their complex differentiation processes result in the unknown effect at different stages. In this study, we focused on the widely used bone forming peptide-1 (BFP-1) and investigated the effect and mechanisms of its addition on the osteogenic induction of hPSCs as a function of the supplementation period.
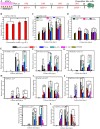
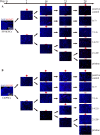
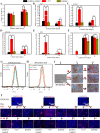
Methods: Monolayer-cultured hPSCs were cultured in osteogenic induction medium for 28 days, and the effect of BFP-1 peptide addition at varying weeks was examined. After differentiation for varying days (0, 7, 14, 21 and 28), the differentiation efficiency was determined by RT-PCR, flow cytometry, immunofluorescence, and alizarin red staining assays. Moreover, the expression of marker genes related to germ layers and epithelial-mesenchymal transition (EMT) was investigated at day 7.
Results: Peptide treatment during the first week promoted the generation of mesoderm cells and mesenchymal-like cells from hiPSCs. Then, the upregulated expression of osteogenesis marker genes/proteins was detected in both hESCs and hiPSCs during subsequent inductions with BFP-1 peptide treatment. Fortunately, further experimental design confirmed that treating the BFP-1 peptide during 7-21 days showed even better performance for hESCs but was ineffective for hiPSCs.
Conclusion: The differentiation efficiency of cells could be improved by determining the optimal treatment period. Our study has great value in maximizing the differentiation of hPSCs by adding osteogenesis peptides based on the revealed mechanisms and promoting the application of hPSCs in bone tissue regeneration.
Keywords: Bone forming peptide; Epithelial-mesenchymal transition; Human pluripotent stem cells; Osteogenic differentiation; Supplement period.
© 2023. Korean Tissue Engineering and Regenerative Medicine Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures





Similar articles
-
In vitro culture and directed osteogenic differentiation of human pluripotent stem cells on peptides-decorated two-dimensional microenvironment.ACS Appl Mater Interfaces. 2015 Mar 4;7(8):4560-72. doi: 10.1021/acsami.5b00188. Epub 2015 Feb 18. ACS Appl Mater Interfaces. 2015. PMID: 25671246
-
Establishing a deeper understanding of the osteogenic differentiation of monolayer cultured human pluripotent stem cells using novel and detailed analyses.Stem Cell Res Ther. 2021 Jan 7;12(1):41. doi: 10.1186/s13287-020-02085-9. Stem Cell Res Ther. 2021. PMID: 33413612 Free PMC article.
-
Investigation of the optimal suspension culture time for the osteoblastic differentiation of human induced pluripotent stem cells using the embryoid body method.Biochem Biophys Res Commun. 2019 Aug 6;515(4):586-592. doi: 10.1016/j.bbrc.2019.05.177. Epub 2019 Jun 6. Biochem Biophys Res Commun. 2019. PMID: 31178132
-
[Mechanism of ring finger protein 11 regulating Akt signaling pathway to promote osteogenic differentiation of bone marrow mesenchymal stem cells].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2022 Jan 15;36(1):102-110. doi: 10.7507/1002-1892.202107108. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2022. PMID: 35038807 Free PMC article. Chinese.
-
Laminins in osteogenic differentiation and pluripotency maintenance.Differentiation. 2020 Jul-Aug;114:13-19. doi: 10.1016/j.diff.2020.05.002. Epub 2020 May 16. Differentiation. 2020. PMID: 32473527 Review.
Cited by
-
Effects of Insulin-like Growth Factor 1 on the Maintenance of Cell Viability and Osteogenic Differentiation of Gingiva-Derived Mesenchymal Stem Cell Spheroids.Medicina (Kaunas). 2025 Jan 4;61(1):76. doi: 10.3390/medicina61010076. Medicina (Kaunas). 2025. PMID: 39859058 Free PMC article.
References
MeSH terms
Substances
Grants and funding
- 2022M721443/China Postdoctoral Science Foundation
- lzujbky-2021-ey14/Fundamental Research Funds for the Central Universities
- lzujbky-2021-kb05/Fundamental Research Funds for the Central Universities
- 20JR5RA150/Natural Science Foundation of Gansu Province
- NO. 21JR7RA863/Natural Science Foundation of Gansu Province
LinkOut - more resources
Full Text Sources
