Self-regulating arousal via pupil-based biofeedback
- PMID: 37904022
- PMCID: PMC10810759
- DOI: 10.1038/s41562-023-01729-z
Self-regulating arousal via pupil-based biofeedback
Abstract
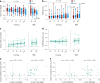
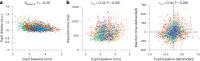
The brain's arousal state is controlled by several neuromodulatory nuclei known to substantially influence cognition and mental well-being. Here we investigate whether human participants can gain volitional control of their arousal state using a pupil-based biofeedback approach. Our approach inverts a mechanism suggested by previous literature that links activity of the locus coeruleus, one of the key regulators of central arousal and pupil dynamics. We show that pupil-based biofeedback enables participants to acquire volitional control of pupil size. Applying pupil self-regulation systematically modulates activity of the locus coeruleus and other brainstem structures involved in arousal control. Furthermore, it modulates cardiovascular measures such as heart rate, and behavioural and psychophysiological responses during an oddball task. We provide evidence that pupil-based biofeedback makes the brain's arousal system accessible to volitional control, a finding that has tremendous potential for translation to behavioural and clinical applications across various domains, including stress-related and anxiety disorders.
© 2023. The Author(s).
Conflict of interest statement
S.N.M., M.B. and N.W. are founders and shareholders of an ETH spin-off called ‘MindMetrix’ that aims to commercialize pupil-based biofeedback, and have a patent application related to the method of pupil-based biofeedback (patent applicant: ETH Zurich; inventors: M.B., S.N.M., N.W., pending patent applications EP21704565.7 and US17/800,455). All other authors declare no competing interests.
Figures
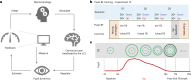
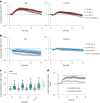
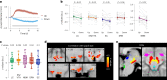
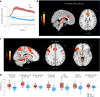

References
MeSH terms
Grants and funding
- CRSK-1_190836/Schweizerischer Nationalfonds zur Förderung der Wissenschaftlichen Forschung (Swiss National Science Foundation)
- 32003B_207719/Schweizerischer Nationalfonds zur Förderung der Wissenschaftlichen Forschung (Swiss National Science Foundation)
- Bridge Discovery (SNF/Innosuisse); 40B2-0_203606/Schweizerischer Nationalfonds zur Förderung der Wissenschaftlichen Forschung (Swiss National Science Foundation)
- Postdoctoral Fellowship Program/Eidgenössische Technische Hochschule Zürich (Federal Institute of Technology Zurich)
- CREATE programme/National Research Foundation Singapore (National Research Foundation-Prime Minister's office, Republic of Singapore)
LinkOut - more resources
Full Text Sources

