CDC Recommendations for Hepatitis C Testing Among Perinatally Exposed Infants and Children - United States, 2023
- PMID: 37906518
- PMCID: PMC10683764
- DOI: 10.15585/mmwr.rr7204a1
CDC Recommendations for Hepatitis C Testing Among Perinatally Exposed Infants and Children - United States, 2023
Abstract
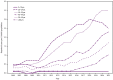
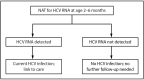
The elimination of hepatitis C is a national priority (https://www.hhs.gov/sites/default/files/Viral-Hepatitis-National-Strategic-Plan-2021-2025.pdf). During 2010-2021, hepatitis C virus (HCV) acute and chronic infections (hereinafter referred to as HCV infections) increased in the United States, consequences of which include cirrhosis, liver cancer, and death. Rates of acute infections more than tripled among reproductive-aged persons during this time (from 0.8 to 2.5 per 100,000 population among persons aged 20-29 years and from 0.6 to 3.5 among persons aged 30-39 years). Because acute HCV infection can lead to chronic infection, this has resulted in increasing rates of HCV infections during pregnancy. Approximately 6%-7% of perinatally exposed (i.e., exposed during pregnancy or delivery) infants and children will acquire HCV infection. Curative direct-acting antiviral therapy is approved by the Food and Drug Administration for persons aged ≥3 years. However, many perinatally infected children are not tested or linked to care. In 2020, because of continued increases in HCV infections in the United States, CDC released universal screening recommendations for adults, which included recommendations for screening for pregnant persons during each pregnancy (Schillie S, Wester C, Osborne M, Wesolowski L, Ryerson AB. CDC recommendations for hepatitis C screening among adults-United States, 2020. MMWR Recomm Rep 2020;69[No. RR-2]:1-17). This report introduces four new CDC recommendations: 1) HCV testing of all perinatally exposed infants with a nucleic acid test (NAT) for detection of HCV RNA at age 2-6 months; 2) consultation with a health care provider with expertise in pediatric hepatitis C management for all infants and children with detectable HCV RNA; 3) perinatally exposed infants and children with an undetectable HCV RNA result at or after age 2 months do not require further follow-up unless clinically warranted; and 4) a NAT for HCV RNA is recommended for perinatally exposed infants and children aged 7-17 months who previously have not been tested, and a hepatitis C virus antibody (anti-HCV) test followed by a reflex NAT for HCV RNA (when anti-HCV is reactive) is recommended for perinatally exposed children aged ≥18 months who previously have not been tested. Proper identification of perinatally infected children, referral to care, and curative treatment are critical to achieving the goal of hepatitis C elimination.
Conflict of interest statement
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflicts of interest were disclosed.
Figures


References
-
- Wilkins T, Malcolm JK, Raina D, Schade RR. Hepatitis C: diagnosis and treatment. Am Fam Physician 2010;81:1351–7. - PubMed
-
- CDC. 2021 Viral hepatitis surveillance report. Atlanta, GA: US Department of Health and Human Services, CDC; 2023. https://www.cdc.gov/hepatitis/statistics/2021surveillance/index.htm
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
