Pyrotinib versus placebo in combination with trastuzumab and docetaxel as first line treatment in patients with HER2 positive metastatic breast cancer (PHILA): randomised, double blind, multicentre, phase 3 trial
- PMID: 37907210
- PMCID: PMC10616786
- DOI: 10.1136/bmj-2023-076065
Pyrotinib versus placebo in combination with trastuzumab and docetaxel as first line treatment in patients with HER2 positive metastatic breast cancer (PHILA): randomised, double blind, multicentre, phase 3 trial
Erratum in
-
Pyrotinib versus placebo in combination with trastuzumab and docetaxel as first line treatment in patients with HER2 positive metastatic breast cancer (PHILA): randomised, double blind, multicentre, phase 3 trial.BMJ. 2023 Nov 16;383:p2665. doi: 10.1136/bmj.p2665. BMJ. 2023. PMID: 37973164 Free PMC article. No abstract available.
Abstract
Objective: To assess the efficacy and safety of pyrotinib (an irreversible pan-HER (human epidermal growth factor receptor) inhibitor), trastuzumab, and docetaxel compared with placebo, trastuzumab, and docetaxel for untreated HER2 positive metastatic breast cancer.
Design: Randomised, double blind, placebo controlled, multicentre, phase 3 trial.
Setting: 40 centres in China between 6 May 2019 and 17 January 2022.
Participants: 590 female patients (median age 52 (interquartile range 46-58) years) with untreated HER2 positive metastatic breast cancer.
Interventions: Eligible patients were randomised 1:1 to receive either oral pyrotinib (400 mg once daily) or placebo, both combined with intravenous trastuzumab (8 mg/kg in cycle 1 and 6 mg/kg in subsequent cycles) and docetaxel (75 mg/m2) on day 1 of each 21 day cycle. Randomisation was stratified by treatment history of trastuzumab in the (neo)adjuvant setting and hormone receptor status. Patients, investigators, and the sponsor's study team were masked to treatment assignment.
Main outcome measures: The primary endpoint was progression-free survival as assessed by the investigator.
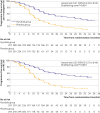
Results: Of the 590 randomised patients, 297 received pyrotinib, trastuzumab, and docetaxel treatment (pyrotinib group), and 293 received placebo, trastuzumab, and docetaxel treatment (placebo group). At data cut-off on 25 May 2022, the median follow-up was 15.5 months. The median progression-free survival according to the investigator was significantly longer in the pyrotinib group than in the placebo group (24.3 (95% confidence interval 19.1 to 33.0) months versus 10.4 (9.3 to 12.3) months; hazard ratio 0.41 (95% confidence interval 0.32 to 0.53); one sided P<0.001). Treatment related adverse events of grade 3 or higher were reported in 267 (90%) of the 297 patients in the pyrotinib group and 224 (76%) of the 293 patients in the placebo group. No treatment related deaths occurred in the pyrotinib group, and one (<1%; diabetic hyperosmolar coma) treatment related death occurred in the placebo group. Survival and toxicities are still under assessment with longer follow-up.
Conclusions: Pyrotinib, trastuzumab, and docetaxel showed superiority by significantly improving progression-free survival compared with placebo, trastuzumab, and docetaxel in patients with untreated HER2 positive metastatic breast cancer. The toxicity was manageable. The findings support this dual anti-HER2 regimen as an alternative first line treatment option in this patient population.
Trial registration: ClinicalTrials.gov NCT03863223.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at https://www.icmje.org/disclosure-of-interest/ and declare: BX has served as advisor or consultant for Novartis and Roche and as a speaker or a member of a speakers’ bureau for AstraZeneca, Pfizer, Roche, and Eisai; JN has received research grants from Hengrui, Sanofi, Qilu, Junshi, Zhengdatianqing, Boan, Maiwei, Novartis, and Xuanzhu; FD, SW, and XZ are employed by Jiangsu Hengrui Pharmaceuticals; no other relationships or activities that could appear to have influenced the submitted work.
Figures


References
-
- Gianni L, Pienkowski T, Im YH, et al. Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trial. Lancet Oncol 2012;13:25-32. 10.1016/S1470-2045(11)70336-9 - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
