Dexmedetomidine Ameliorates Cardiac Ischemia/Reperfusion Injury by Enhancing Autophagy Through Activation of the AMPK/SIRT3 Pathway
- PMID: 37908314
- PMCID: PMC10613569
- DOI: 10.2147/DDDT.S428024
Dexmedetomidine Ameliorates Cardiac Ischemia/Reperfusion Injury by Enhancing Autophagy Through Activation of the AMPK/SIRT3 Pathway
Abstract
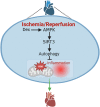
Objective: Myocardial ischemia-reperfusion (I/R) injury is a detrimental disease, resulting in high morbidity and mortality globally. In this study, we aimed to investigate the role of Dex in mitigating cardiac I/R injury.
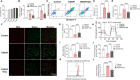
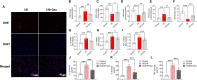
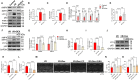
Methods: H9c2 cells were treated with Dex (1 μM) for 24 h followed by oxygen-glucose deprivation/re-oxygenation (OGD/R). ANP and BNP mRNA of H9c2 cells and the LDH release were measured. Apoptosis of H9c2 cells was analyzed by flow cytometry. Mitochondrial membrane potential and superoxide production were detected by JC-1 staining and MitoSOXTM Red, respectively. Cell aerobic respiration was measured using Seahorse analysis. In vivo, mice were injected with Dex (25 μg/kg, i.p., once daily) for 5 days and then subjected to heart I/R. Heart function was analyzed by echocardiography. CK-MB and LDH were measured by Elisa. Infarct size was measured using TTC-Evans blue staining. Mitochondrial ultrastructure was observed using transmission electron microscopy. DHE staining, SOD activity, the content of MDA, and the content of GSH/GSSG of heart were measured to evaluate the oxidative stress. In addition, inflammatory cytokines were measured in vivo and in vitro. Furthermore, AMPK, SIRT3, and autophagy-related protein expression in the heart were detected by Western blot.
Results: Dex reduced the H9c2 cells injury exposed to OGD/R, accompanied by improved mitochondrial function and membrane potential. In vivo, Dex improved heart function, myocardial injury, and the mitochondria ultrastructure following I/R injury. Meanwhile, Dex inhibited myocardial oxidative stress and inflammation in the myocardial I/R. Furthermore, Compound C (an AMPK inhibitor) could inhibit Dex-induced autophagy in the I/R heart and the 3-MA (an autophagy inhibitor) could partially interfere with the effects of Dex on the protection of I/R heart.
Conclusion: Dex suppressed oxidative stress and inflammation by promoting autophagy through activating the AMPK/SIRT3 pathway, thus protecting the heart against the I/R injury.
Keywords: AMPK/SIRT3; autophagy; cardiac ischemia reperfusion; dexmedetomidine.
© 2023 He et al.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures

Similar articles
-
Dexmedetomidine Confers Protection Against Neuronal Oxygen Glucose Deprivation-Reperfusion by Regulating SIRT3 Mediated Autophagy.Neurochem Res. 2022 Nov;47(11):3490-3505. doi: 10.1007/s11064-022-03712-y. Epub 2022 Aug 30. Neurochem Res. 2022. PMID: 36042140
-
Dexmedetomidine postconditioning attenuates myocardial ischemia/reperfusion injury by activating the Nrf2/Sirt3/SOD2 signaling pathway in the rats.Redox Rep. 2023 Dec;28(1):2158526. doi: 10.1080/13510002.2022.2158526. Redox Rep. 2023. PMID: 36738240 Free PMC article.
-
The Role of the Sirt1/Foxo3a Pathway in Mitigating Myocardial Ischemia-Reperfusion Injury by Dexmedetomidine.Chem Biol Drug Des. 2025 Apr;105(4):e70100. doi: 10.1111/cbdd.70100. Chem Biol Drug Des. 2025. PMID: 40230274 Free PMC article.
-
Perioperative acute kidney injury: The renoprotective effect and mechanism of dexmedetomidine.Biochem Biophys Res Commun. 2024 Feb 5;695:149402. doi: 10.1016/j.bbrc.2023.149402. Epub 2023 Dec 23. Biochem Biophys Res Commun. 2024. PMID: 38159412 Review.
-
Dexmedetomidine as a cardioprotective drug: a narrative review.J Anesth. 2023 Dec;37(6):961-970. doi: 10.1007/s00540-023-03261-w. Epub 2023 Sep 26. J Anesth. 2023. PMID: 37750978 Review.
Cited by
-
Dexmedetomidine alleviates intestinal ischemia/reperfusion injury by modulating intestinal neuron autophagy and mitochondrial homeostasis via Nupr1 regulation.Mol Med. 2024 Nov 6;30(1):203. doi: 10.1186/s10020-024-00952-2. Mol Med. 2024. PMID: 39508252 Free PMC article.
-
Cardioprotective strategies in myocardial ischemia-reperfusion injury: Implications for improving clinical translation.J Mol Cell Cardiol Plus. 2024 Dec 16;11:100278. doi: 10.1016/j.jmccpl.2024.100278. eCollection 2025 Mar. J Mol Cell Cardiol Plus. 2024. PMID: 40182153 Free PMC article. Review.
-
AAV-mediated STC-1 expression mitigates neuroinflammation and preserves visual function in degenerative retinopathy.J Transl Med. 2025 Aug 18;23(1):924. doi: 10.1186/s12967-025-06898-1. J Transl Med. 2025. PMID: 40826460 Free PMC article.
-
Dexmedetomidine reduces acute lung injury caused by LPS through the SIRT3 signaling pathway in vivo.Front Pharmacol. 2025 Jun 25;16:1524219. doi: 10.3389/fphar.2025.1524219. eCollection 2025. Front Pharmacol. 2025. PMID: 40635757 Free PMC article.
-
Dexmedetomidine improves clinical outcomes in sepsis-induced myocardial injury: a retrospective cohort study.Front Pharmacol. 2025 Jan 15;15:1529167. doi: 10.3389/fphar.2024.1529167. eCollection 2024. Front Pharmacol. 2025. PMID: 39881865 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

