Peptide-drug co-assembling: A potent armament against cancer
- PMID: 37908727
- PMCID: PMC10614680
- DOI: 10.7150/thno.87356
Peptide-drug co-assembling: A potent armament against cancer
Abstract
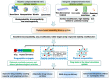
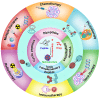
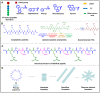
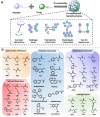
Cancer is still one of the major problems threatening human health and the therapeutical efficacies of available treatment choices are often rather low. Due to their favorable biocompatibility, simplicity of modification, and improved therapeutic efficacy, peptide-based self-assembled delivery systems have undergone significant evolution. Physical encapsulation and covalent conjugation are two common approaches to load drugs for peptide assembly-based delivery, which are always associated with drug leaks in the blood circulation system or changed pharmacological activities, respectively. To overcome these difficulties, a more elegant peptide-based assembly strategy is desired. Notably, peptide-mediated co-assembly with drug molecules provides a new method for constructing nanomaterials with improved versatility and structural stability. The co-assembly strategy can be used to design various nanostructures for cancer therapy, such as nanotubes, nanofibrils, hydrogels, and nanovesicles. Recently, these co-assembled nanostructures have gained tremendous attention for their unique superiorities in tumor therapy. This article describes the classification of assembled peptides, driving forces for co-assembly, and specifically, the design methodologies for various drug molecules in co-assembly. It also highlights recent research on peptide-mediated co-assembled delivery systems for cancer therapy. Finally, it summarizes the pros and cons of co-assembly in cancer therapy and offers some suggestions for conquering the challenges in this field.
Keywords: cancer therapy; co-assembly; drug delivery; nanostructures; peptide.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures




Similar articles
-
Recent advances in self-assembled peptides: Implications for targeted drug delivery and vaccine engineering.Adv Drug Deliv Rev. 2017 Feb;110-111:169-187. doi: 10.1016/j.addr.2016.06.013. Epub 2016 Jun 26. Adv Drug Deliv Rev. 2017. PMID: 27356149 Review.
-
Self-Assembled Peptide Drug Delivery Systems.ACS Appl Bio Mater. 2021 Jan 18;4(1):24-46. doi: 10.1021/acsabm.0c00707. Epub 2020 Jul 27. ACS Appl Bio Mater. 2021. PMID: 35014275 Review.
-
Peptide Nanomaterials for Drug Delivery Applications.Curr Protein Pept Sci. 2020;21(4):401-412. doi: 10.2174/1389203721666200101091834. Curr Protein Pept Sci. 2020. PMID: 31893991 Review.
-
Peptide-Based Nanoassemblies in Gene Therapy and Diagnosis: Paving the Way for Clinical Application.Molecules. 2020 Jul 31;25(15):3482. doi: 10.3390/molecules25153482. Molecules. 2020. PMID: 32751865 Free PMC article. Review.
-
Self-assembling peptides-based nano-cargos for targeted chemotherapy and immunotherapy of tumors: recent developments, challenges, and future perspectives.Drug Deliv. 2022 Dec;29(1):1184-1200. doi: 10.1080/10717544.2022.2058647. Drug Deliv. 2022. PMID: 35403517 Free PMC article. Review.
Cited by
-
Evaluating TcAs for Use in Biotechnology Applications.BioTech (Basel). 2025 Jan 25;14(1):5. doi: 10.3390/biotech14010005. BioTech (Basel). 2025. PMID: 39982272 Free PMC article.
-
Current progress and remaining challenges of peptide-drug conjugates (PDCs): next generation of antibody-drug conjugates (ADCs)?J Nanobiotechnology. 2025 Apr 22;23(1):305. doi: 10.1186/s12951-025-03277-2. J Nanobiotechnology. 2025. PMID: 40259322 Free PMC article. Review.
-
Peptide-Drug Conjugates: A New Hope for Cancer.J Pept Sci. 2025 Aug;31(8):e70040. doi: 10.1002/psc.70040. J Pept Sci. 2025. PMID: 40646707 Free PMC article. Review.
-
Trypsin-instructed bioactive peptide nanodrugs with cascading transformations to improve chemotherapy against colon cancer.J Nanobiotechnology. 2025 Jan 31;23(1):66. doi: 10.1186/s12951-025-03143-1. J Nanobiotechnology. 2025. PMID: 39891144 Free PMC article.
-
Recent Advances in Smart Linkage Strategies for Developing Drug Conjugates for Targeted Delivery.Top Curr Chem (Cham). 2025 Mar 13;383(2):13. doi: 10.1007/s41061-025-00497-w. Top Curr Chem (Cham). 2025. PMID: 40080285 Review.
References
-
- Byrd DR, Brierley JD, Baker TP, Sullivan DC, Gress DM. Current and future cancer staging after neoadjuvant treatment for solid tumors. CA Cancer J Clin. 2021;71:140–8. - PubMed
-
- Youn YS, Bae YH. Perspectives on the past, present, and future of cancer nanomedicine. Adv Drug Deliv Rev. 2018;130:3–11. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

