Efficacy and safety of adalimumab biosimilar (HS016) in inflammatory bowel disease from the real-world study
- PMID: 37908975
- PMCID: PMC10613675
- DOI: 10.3389/fphar.2023.1259183
Efficacy and safety of adalimumab biosimilar (HS016) in inflammatory bowel disease from the real-world study
Abstract
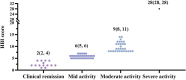
Objective: Adalimumab (ADA) is an effective treatment for inflammatory bowel disease (IBD), both ulcerative colitis (UC) and Crohn's disease (CD). The equal effect between the original ADA and biosimilars from Europe and the United States has been shown. However, the biosimilar of ADA is different in China. The effectiveness and safety data of ADA biosimilar (HS016) in China have yet to be discovered. Patients and methods: 91 patients (75 CD, 16 UC) received HS016 treatment and were enrolled in this study. Therapeutic response and safety profiles were analyzed. Therapeutic drug monitoring (TDM) was also carried out among nonresponse patients. After being considered as "nonresponse" (after three or 6 months of treatment), 20 patients' serum TNFα concentrations were measured and correlated to their disease severity. Results: Among active CD patients (n = 61), 75.4% (46/61) at 12 w, 73.8% (45/61) at 26 w, 50.8% (31/61) at 52 w achieved the clinical response, respectively; 55.7% (34/61) at 12 w, 65.6% (40/61) at 26 w, and 45.9% (28/61) at 52 w achieved clinical remission. The maintained remission rates of CD (n = 14) in clinical remission were 100% (14/14) at 12 w, 78.6% (11/14) at 26 w, and 63.6% (7/11) at 52 w, respectively. Among active UC patients, 37.5% (6/16) at 12 w and 50% (8/16) at 26 w achieved clinical response. Total adverse event rates were 5.5% (5/91) during 52-week visits. Due to the inadequate serum drug concentration, 30.4% (7/23) of patients had poor clinical responses. Elevations of serum anti-drug antibodies occurred in one additional patient (4.3%). Conclusion: ADA biosimilar HS016 had good efficacy and safety in Chinese IBD patients.
Keywords: Crohn’s disease; adalimumab biosimilar; therapeutic drug monitoring; therapeutic effect evaluation; ulcerative colitis.
Copyright © 2023 Wang, Li, Shi, Zhou, Yang, Li, Wu and Liang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Amini Kadijani A., Asadzadeh Aghdaei H., Sorrentino D., Mirzaei A., Shahrokh S., Balaii H., et al. (2017). Transmembrane TNF-α density, but not soluble TNF-α level, is associated with primary response to infliximab in inflammatory bowel disease. Clin. Transl. Gastroenterol. 8 (9), e117. 10.1038/ctg.2017.44 - DOI - PMC - PubMed
-
- Billiet T., Cleynen I., Ballet V., Claes K., Princen F., Singh S., et al. (2017). Evolution of cytokines and inflammatory biomarkers during infliximab induction therapy and the impact of inflammatory burden on primary response in patients with Crohn's disease. Scand. J. Gastroenterol. 52 (10), 1086–1092. 10.1080/00365521.2017.1339825 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

