Inhibition of polyamine biosynthesis preserves β cell function in type 1 diabetes
- PMID: 37918404
- PMCID: PMC10694631
- DOI: 10.1016/j.xcrm.2023.101261
Inhibition of polyamine biosynthesis preserves β cell function in type 1 diabetes
Abstract
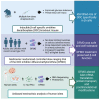
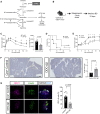
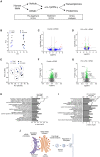
In preclinical models, α-difluoromethylornithine (DFMO), an ornithine decarboxylase (ODC) inhibitor, delays the onset of type 1 diabetes (T1D) by reducing β cell stress. However, the mechanism of DFMO action and its human tolerability remain unclear. In this study, we show that mice with β cell ODC deletion are protected against toxin-induced diabetes, suggesting a cell-autonomous role of ODC during β cell stress. In a randomized controlled trial (ClinicalTrials.gov: NCT02384889) involving 41 recent-onset T1D subjects (3:1 drug:placebo) over a 3-month treatment period with a 3-month follow-up, DFMO (125-1,000 mg/m2) is shown to meet its primary outcome of safety and tolerability. DFMO dose-dependently reduces urinary putrescine levels and, at higher doses, preserves C-peptide area under the curve without apparent immunomodulation. Transcriptomics and proteomics of DFMO-treated human islets exposed to cytokine stress reveal alterations in mRNA translation, nascent protein transport, and protein secretion. These findings suggest that DFMO may preserve β cell function in T1D through islet cell-autonomous effects.
Keywords: disease modification; islet; ornithine decarboxylase; polyamines; prevention; trial; type 1 diabetes; α-difluoromethylornithine; β cell; β cell stress.
Copyright © 2023 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests R.G.M., L.A.D., and E.W.G. are coauthors on a patent application using DFMO for treatment of β cell dysfunction in T1D. E.W.G. is an employee of Cancer Prevention Pharmaceuticals.
Figures

References
-
- Foster N.C., Beck R.W., Miller K.M., Clements M.A., Rickels M.R., DiMeglio L.A., Maahs D.M., Tamborlane W.V., Bergenstal R., Smith E., et al. State of Type 1 Diabetes Management and Outcomes from the T1D Exchange in 2016-2018. Diabetes Technol. Therapeut. 2019;21:66–72. doi: 10.1089/dia.2018.0384. - DOI - PMC - PubMed
-
- Rawshani A., Sattar N., Franzén S., Rawshani A., Hattersley A.T., Svensson A.M., Eliasson B., Gudbjörnsdottir S. Excess mortality and cardiovascular disease in young adults with type 1 diabetes in relation to age at onset: a nationwide, register-based cohort study. Lancet. 2018;392:477–486. doi: 10.1016/S0140-6736(18)31506-X. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- UL1 TR002529/TR/NCATS NIH HHS/United States
- I01 BX001733/BX/BLRD VA/United States
- R01 DK127236/DK/NIDDK NIH HHS/United States
- P30 DK097512/DK/NIDDK NIH HHS/United States
- R01 DK121987/DK/NIDDK NIH HHS/United States
- R01 DK124906/DK/NIDDK NIH HHS/United States
- P30 DK020595/DK/NIDDK NIH HHS/United States
- R01 DK133881/DK/NIDDK NIH HHS/United States
- R01 DK121929/DK/NIDDK NIH HHS/United States
- R01 DK127308/DK/NIDDK NIH HHS/United States
- T32 AI153020/AI/NIAID NIH HHS/United States
- U01 DK127786/DK/NIDDK NIH HHS/United States
- R01 DK060581/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

