MDA-9/Syntenin in the tumor and microenvironment defines prostate cancer bone metastasis
- PMID: 37922327
- PMCID: PMC10636346
- DOI: 10.1073/pnas.2307094120
MDA-9/Syntenin in the tumor and microenvironment defines prostate cancer bone metastasis
Abstract
Bone metastasis is a frequent and incurable consequence of advanced prostate cancer (PC). An interplay between disseminated tumor cells and heterogeneous bone resident cells in the metastatic niche initiates this process. Melanoma differentiation associated gene-9 (mda-9/Syntenin/syndecan binding protein) is a prometastatic gene expressed in multiple organs, including bone marrow-derived mesenchymal stromal cells (BM-MSCs), under both physiological and pathological conditions. We demonstrate that PDGF-AA secreted by tumor cells induces CXCL5 expression in BM-MSCs by suppressing MDA-9-dependent YAP/MST signaling. CXCL5-derived tumor cell proliferation and immune suppression are consequences of the MDA-9/CXCL5 signaling axis, promoting PC disease progression. mda-9 knockout tumor cells express less PDGF-AA and do not develop bone metastases. Our data document a previously undefined role of MDA-9/Syntenin in the tumor and microenvironment in regulating PC bone metastasis. This study provides a framework for translational strategies to ameliorate health complications and morbidity associated with advanced PC.
Keywords: CXCL5; MDA-9/Syntenin; PDGF-AA; bone marrow–derived mesenchymal stromal cell; bone metastasis.
Conflict of interest statement
The authors declare no competing interest.
Figures



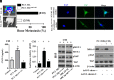
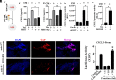

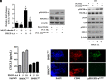


References
-
- Rigaud J., et al. , Prognostic value of bone scan in patients with metastatic prostate cancer treated initially with androgen deprivation therapy. J. Urol. 168, 1423–1426 (2002). - PubMed
-
- Ye L., Kynaston H. G., Jiang W. G., Bone metastasis in prostate cancer: Molecular and cellular mechanisms (Review). Int. J. Mol. Med. 20, 103–111 (2007). - PubMed
-
- Coghlin C., Murray G. I., Current and emerging concepts in tumour metastasis. J. Pathol. 222, 1–15 (2010). - PubMed
-
- Pneumaticos S. G., et al. , Osteoprotegerin expression during the micro- and macrometastatic phases of the osteoblastic metastasis in prostate cancer: Therapeutic implications. Expert Opin. Ther Targets 17, 1395–1403 (2013). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

