The state of cell and gene therapy in 2023
- PMID: 37927037
- PMCID: PMC10727993
- DOI: 10.1016/j.ymthe.2023.11.001
The state of cell and gene therapy in 2023
Abstract
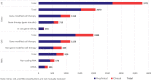
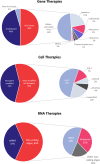
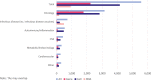
Progress in the understanding of human diseases has coincided with the advent of precision medicine, whereby the underlying genetic and molecular contributors can be used as diagnostic and therapeutic biomarkers. To address these, drug developers have designed a range of different treatment strategies, including gene therapy, which the American Society of Gene and Cell Therapy defines as the use of genetic material to treat or prevent disease. A number of approaches exist, including the delivery of genetic material in vivo or ex vivo, as well as the use of RNA species to alter gene expression in particular disease states. Through the end of the first quarter of 2023, there were more than 100 different approved gene, cell, and RNA therapies throughout the world, with over 3,700 more in clinical and preclinical development. This review comprehensively captures the landscape for such advanced therapies, including the different genetic technologies used and diseases targeted in clinical trials.
Keywords: RNA therapy; approved products; cell therapy; clinical trials; gene therapy; genetically modified cell therapy.
Copyright © 2023. Published by Elsevier Inc.
Conflict of interest statement
Declaration of interests D.B. is an employee of ASGCT. S.M., D.C., and F.E. are employees of Citeline. L.N.-J. is a former employee of Citeline. ASGCT and Citeline are partners in the development of the underlying data, which is derived from Citeline products and made available in the Gene, Cell & RNA Therapy Landscape quarterly data report.
Figures



References
-
- Goodkey K., Aslesh T., Maruyama R., Yokota T. Nusinersen in the Treatment of Spinal Muscular Atrophy. Methods Mol. Biol. 2018;1828:69–76. - PubMed
-
- Hoy S.M. Onasemnogene Abeparvovec: First Global Approval. Drugs. 2019;79:1255–1262. - PubMed
-
- Novartis . 2023. Novartis Shares Zolgensma Long-Term Data Demonstrating Sustained Durability up to 7.5 Years Post-dosing; 100% Achievement of All Assessed Milestones in Children Treated Prior to SMA Symptom Onset.https://www.novartis.com/news/media-releases/novartis-shares-zolgensma-l...
-
- Boyiadzis M.M., Dhodapkar M.V., Brentjens R.J., Kochenderfer J.N., Neelapu S.S., Maus M.V., Porter D.L., Maloney D.G., Grupp S.A., Mackall C.L., et al. Chimeric antigen receptor (CAR) T therapies for the treatment of hematologic malignancies: clinical perspective and significance. J. Immunother. Cancer. 2018;6:137. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

