Pilot randomized controlled trial of restricted versus liberal crystalloid fluid management in pediatric post-operative and trauma patients
- PMID: 37941073
- PMCID: PMC10631167
- DOI: 10.1186/s40814-023-01408-w
Pilot randomized controlled trial of restricted versus liberal crystalloid fluid management in pediatric post-operative and trauma patients
Abstract
Background: Intravenous (IV) fluid therapy is essential in the treatment of critically ill pediatric surgery and trauma patients. Recent studies have suggested that aggressive fluids may be detrimental to patients. Prospective studies are needed to compare liberal to restricted fluid management in these patients. The primary objective of this pilot trial is to test study feasibility-recruitment and adherence to the study treatment algorithm.
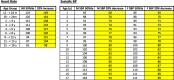
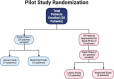
Methods: We conducted a two-part pilot randomized controlled trial (RCT) comparing liberal to restricted crystalloid fluid management in 50 pediatric post-operative (1-18 years) and trauma (1-15 years) patients admitted to our pediatric intensive care unit (PICU). Patients were randomized to a high (liberal) volume or low (restricted) volume algorithm using unblinded, blocked randomization. A revised treatment algorithm was used after the 29th patient for the second part of the RCT. The goal of the trial was to determine the feasibility of conducting an RCT at a single site for recruitment and retention. We also collected data on the safety of study interventions and clinical outcomes, including pulmonary, infectious, renal, post-operative, and length of stay outcomes.
Results: Fifty patients were randomized to either liberal (n = 26) or restricted (n = 24) fluid management strategy. After data was obtained on 29 patients, a first study analysis was performed. The volume of fluid administered and triggers for intervention were adapted to optimize the treatment effect and clarity of outcomes. Updated and refined fluid management algorithms were created. These were used for the second part of the RCT on patients 30-50. During this second study period, 54% (21/39, 95% CI 37-70%) of patients approached were enrolled in the study. Of the patients enrolled, 71% (15/21, 95% CI 48-89%) completed the study. This met our a priori recruitment and retention criteria for success. A data safety monitoring committee concluded that no adverse events were related to study interventions. Although the study was not powered to detect differences in outcomes, after the algorithm was revised, we observed a non-significant trend towards improved pulmonary outcomes in patients on the restricted arm, including decreased need for and time on oxygen support and decreased need for mechanical ventilation.
Conclusion: We demonstrated the feasibility and safety of conducting a single-site RCT comparing liberal to restricted crystalloid fluid management in critically ill pediatric post-operative and trauma patients. We observed trends in improved pulmonary outcomes in patients undergoing restricted fluid management. A definitive multicenter RCT comparing fluid management strategies in these patients is warranted.
Trial registration: ClinicalTrials.gov, NCT04201704 . Registered 17 December 2019-retrospectively registered.
Keywords: Feasibility; Fluid management; Fluid resuscitation; Pediatric trauma; Randomized controlled trial.
© 2023. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures






References
-
- Xu J, Murphy SL, Kochanek KD, Bastian B, Arias E. Deaths: Final Data for 2016. US Department Of Health And Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics,National Vital Statistics System. 2018;67(5):1–75. - PubMed
-
- Kochanek KD, Murphy SL, Xu J, Arias E. Deaths: Final Data for 2017. US department of health and human services, Centers for Disease Control and Prevention, National Center for Health Statistic. Nat Vital Statis Syst. 2019;68(9):1–76. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

