Current Advances on Nanomaterials Interfering with Lactate Metabolism for Tumor Therapy
- PMID: 37941489
- PMCID: PMC10797484
- DOI: 10.1002/advs.202305662
Current Advances on Nanomaterials Interfering with Lactate Metabolism for Tumor Therapy
Abstract
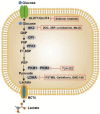
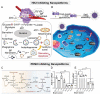
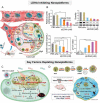
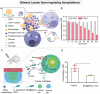
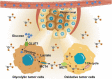
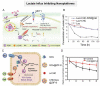
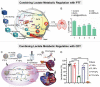
Increasing numbers of studies have shown that tumor cells prefer fermentative glycolysis over oxidative phosphorylation to provide a vast amount of energy for fast proliferation even under oxygen-sufficient conditions. This metabolic alteration not only favors tumor cell progression and metastasis but also increases lactate accumulation in solid tumors. In addition to serving as a byproduct of glycolytic tumor cells, lactate also plays a central role in the construction of acidic and immunosuppressive tumor microenvironment, resulting in therapeutic tolerance. Recently, targeted drug delivery and inherent therapeutic properties of nanomaterials have attracted great attention, and research on modulating lactate metabolism based on nanomaterials to enhance antitumor therapy has exploded. In this review, the advanced tumor therapy strategies based on nanomaterials that interfere with lactate metabolism are discussed, including inhibiting lactate anabolism, promoting lactate catabolism, and disrupting the "lactate shuttle". Furthermore, recent advances in combining lactate metabolism modulation with other therapies, including chemotherapy, immunotherapy, photothermal therapy, and reactive oxygen species-related therapies, etc., which have achieved cooperatively enhanced therapeutic outcomes, are summarized. Finally, foreseeable challenges and prospective developments are also reviewed for the future development of this field.
Keywords: glycolysis; lactate metabolism; nanoparticles; tumor therapy.
© 2023 The Authors. Advanced Science published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures








References
-
- a) Begg A. C., Stewart F. A., Vens C., Nat. Rev. Cancer 2011, 11, 239; - PubMed
- b) Martin J. D., Cabral H., Stylianopoulos T., Jain R. K., Nat. Rev. Clin. Oncol. 2020, 17, 251; - PMC - PubMed
- c) McCormick F., Nat. Rev. Cancer 2001, 1, 130; - PubMed
- d) Liu Y., Bhattarai P., Dai Z., Chen X., Chem. Soc. Rev. 2019, 48, 2053; - PMC - PubMed
- e) Pham T. C., Nguyen V. N., Choi Y., Lee S., Yoon J., Chem. Rev. 2021, 121, 13454; - PubMed
- f) Guo W., Chen Z., Tan L., Gu D., Ren X., Fu C., Wu Q., Meng X., View 2021, 3, 20200174.
-
- Sun Y., Cancer Lett. 2016, 380, 205. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
