Natural history of Duchenne muscular dystrophy in the United Kingdom: A descriptive study using the Clinical Practice Research Datalink
- PMID: 37957895
- PMCID: PMC10726817
- DOI: 10.1002/brb3.3331
Natural history of Duchenne muscular dystrophy in the United Kingdom: A descriptive study using the Clinical Practice Research Datalink
Abstract
Background: Duchenne muscular dystrophy (DMD) is a rare, muscle-degenerative disease predominantly affecting males. Natural history models capture the full disease pathway under current care and combine with estimates of new interventions' effects to assess cost-effectiveness by health technology decision-makers. These models require mortality estimates throughout a patient's lifetime, but rare disease datasets typically contain relatively few patients with short follow-ups. Alternative (published) sources of mortality data may therefore be required.
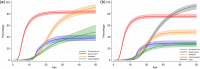
Methods: The Clinical Practice Research Datalink (CPRD) was evaluated as a source of mortality and natural history data for future economic evaluations of health technologies for DMD and rare diseases in general in the UK population. This retrospective longitudinal cohort study provides flexible parametric estimates of mortality rates and survival probabilities in the current UK DMD population through primary/secondary records in the CPRD since 1990. It also investigates clinically significant milestones such as corticosteroid use, spinal surgery, and cardiomyopathy in these patients.
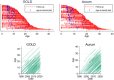
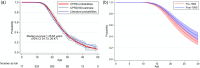
Results: A total of 1121 male patients were included in the study, observed from 0.7 to 48.9 years. Median life expectancy was 25.64 years (95% confidence interval 24.73, 26.47), consistent with previous global estimates. This has improved to 26.47 (25.16, 27.89) years in patients born after 1990. The median ages at corticosteroid initiation, spinal surgery, ventilation, and cardiomyopathy diagnosis were 6.06 years (5.77, 6.29), 14.79 years (14.29, 15.09), 16.97 years (16.50, 18.31), and 15.26 years (14.22, 16.70), respectively.
Conclusions: Estimates of mortality in UK-based DMD patients are age-specific in a uniquely large and nationally representative sample from the CPRD.
Keywords: Duchenne muscular dystrophy; economic models; electronic health records; statistical models.
© 2023 The Authors. Brain and Behavior published by Wiley Periodicals LLC.
Conflict of interest statement
Jonathan Broomfield, Keith Abrams and Michael Crowther have received consultancy fees from Duchenne UK. Michela Guglieri has received research funding from Sarepta, PTC, Muscular Dystrophy UK, NIH and H2020; is (or has been) chief investigator/principal investigator for clinical trials of Pfizer, Italfarmaco, Santhera, Roche, ReveraGen, Dynacure, Dyne; is a member of Advisory boards for Pfizer, NS Pharma, Dyne (honoraria through Newcastle University); and has received speaker honoraria from Sarepta, Italfarmaco, Novartis and Roche.
Figures


References
-
- Birnkrant, D. J. , Bello, L. , Butterfird, R. J. , Carter, J. C. , Cripe, L. H. , Cripe, T. P. , McKim, D. A. , Nandi, D. , & Pegoraro, E. (2022). Cardiorespiratory management of Duchenne muscular dystrophy: Emerging therapies, neuromuscular genetics, and new clinical challenges. Lancet Respiratory Medicine, 10(4), 403–420. - PubMed
-
- Birnkrant, D. J. , Bushby, K. , Bann, C. M. , Apkon, S. D. , Blackwell, A. , Brumbaugh, D. , Case, L. E. , Clemens, P. R. , Hadjiyannakis, S. , Pandya, S. , Street, N. , Tomezsko, J. , Wagner, K. R. , Ward, L. M. , Weber, D. R. , & DMD Care Considerations Working Group . (2018). Diagnosis and management of Duchenne muscular dystrophy, part 1: Diagnosis, and neuromuscular, rehabilitation, endocrine, and gastrointestinal and nutritional management. Lancet Neurology, 17(3), 251–267. - PMC - PubMed
-
- Broomfield, J. , Hill, M. , Chandler, F. , Crowther, M. , Godfrey, J. , Guglieri, M. , Hastie, J. , Larkindale, J. , Mumby‐Croft, J. , Reuben, E. , Woodcock, F. , & Abrams, K. (2023). Developing a natural history model for Duchenne muscular dystrophy. Research Square, 10.21203/rs.3.rs-2405860/v1 - DOI - PMC - PubMed
-
- Bushby, K. , Finkel, F. , Birnkrant, D. J. , Case, L. E. , Clemens, P. R. , Cripe, L. , Kaul, A. , Kinnett, K. , McDonald, C. , Pandya, S. , Poysky, J. , Shapiro, F. , Tomezsko, J. , Constantin, C. , & DMD Care Considerations Working Group . (2010a). Diagnosis and management of Duchenne Muscular Dystrophy, part 1: Diagnosis, and pharmacological and psychosocial management. Lancet Neurology, 9, 77–93. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

