Missense mutations in CRX homeodomain cause dominant retinopathies through two distinct mechanisms
- PMID: 37963072
- PMCID: PMC10645426
- DOI: 10.7554/eLife.87147
Missense mutations in CRX homeodomain cause dominant retinopathies through two distinct mechanisms
Abstract
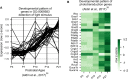
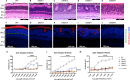
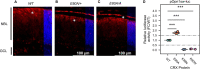
Homeodomain transcription factors (HD TFs) are instrumental to vertebrate development. Mutations in HD TFs have been linked to human diseases, but their pathogenic mechanisms remain elusive. Here, we use Cone-Rod Homeobox (CRX) as a model to decipher the disease-causing mechanisms of two HD mutations, p.E80A and p.K88N, that produce severe dominant retinopathies. Through integrated analysis of molecular and functional evidence in vitro and in knock-in mouse models, we uncover two novel gain-of-function mechanisms: p.E80A increases CRX-mediated transactivation of canonical CRX target genes in developing photoreceptors; p.K88N alters CRX DNA-binding specificity resulting in binding at ectopic sites and severe perturbation of CRX target gene expression. Both mechanisms produce novel retinal morphological defects and hinder photoreceptor maturation distinct from loss-of-function models. This study reveals the distinct roles of E80 and K88 residues in CRX HD regulatory functions and emphasizes the importance of transcriptional precision in normal development.
Keywords: CRX disease mutations; DNA-binding specificity; genetics; genomics; homeodomain; inherited retinal diseases; mouse; neuroscience; photoreceptor development; transcription factor.
© 2023, Zheng et al.
Conflict of interest statement
YZ, CS, XZ, PR, SC No competing interests declared
Figures





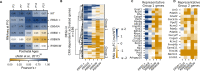





Update of
-
Missense mutations in CRX homeodomain cause dominant retinopathies through two distinct mechanisms.bioRxiv [Preprint]. 2023 Jun 15:2023.02.01.526652. doi: 10.1101/2023.02.01.526652. bioRxiv. 2023. Update in: Elife. 2023 Nov 14;12:RP87147. doi: 10.7554/eLife.87147. PMID: 36778408 Free PMC article. Updated. Preprint.
Similar articles
-
Aberrant homeodomain-DNA cooperative dimerization underlies distinct developmental defects in two dominant CRX retinopathy models.bioRxiv [Preprint]. 2024 Mar 14:2024.03.12.584677. doi: 10.1101/2024.03.12.584677. bioRxiv. 2024. Update in: Genome Res. 2025 Feb 14;35(2):242-256. doi: 10.1101/gr.279340.124. PMID: 38559186 Free PMC article. Updated. Preprint.
-
Aberrant homeodomain-DNA cooperative dimerization underlies distinct developmental defects in two dominant CRX retinopathy models.Genome Res. 2025 Feb 14;35(2):242-256. doi: 10.1101/gr.279340.124. Genome Res. 2025. PMID: 39715683 Free PMC article.
-
Missense mutations in CRX homeodomain cause dominant retinopathies through two distinct mechanisms.bioRxiv [Preprint]. 2023 Jun 15:2023.02.01.526652. doi: 10.1101/2023.02.01.526652. bioRxiv. 2023. Update in: Elife. 2023 Nov 14;12:RP87147. doi: 10.7554/eLife.87147. PMID: 36778408 Free PMC article. Updated. Preprint.
-
Mechanisms of blindness: animal models provide insight into distinct CRX-associated retinopathies.Dev Dyn. 2014 Oct;243(10):1153-66. doi: 10.1002/dvdy.24151. Epub 2014 Jun 27. Dev Dyn. 2014. PMID: 24888636 Free PMC article. Review.
-
Cone Rod Homeobox (CRX): literature review and new insights.Ophthalmic Genet. 2025 Aug;46(4):338-346. doi: 10.1080/13816810.2025.2458086. Epub 2025 Mar 12. Ophthalmic Genet. 2025. PMID: 40074530 Review.
Cited by
-
Active DNA demethylation upstream of rod-photoreceptor fate determination is required for retinal development.PLoS Biol. 2025 Aug 4;23(8):e3003332. doi: 10.1371/journal.pbio.3003332. eCollection 2025 Aug. PLoS Biol. 2025. PMID: 40758714 Free PMC article.
-
Transcriptional precision in photoreceptor development and diseases - Lessons from 25 years of CRX research.Front Cell Neurosci. 2024 Feb 13;18:1347436. doi: 10.3389/fncel.2024.1347436. eCollection 2024. Front Cell Neurosci. 2024. PMID: 38414750 Free PMC article. Review.
-
Pathogenic variants in Crx have distinct cis-regulatory effects on enhancers and silencers in photoreceptors.bioRxiv [Preprint]. 2023 Dec 2:2023.05.27.542576. doi: 10.1101/2023.05.27.542576. bioRxiv. 2023. Update in: Genome Res. 2024 Mar 20;34(2):243-255. doi: 10.1101/gr.278133.123. PMID: 37292699 Free PMC article. Updated. Preprint.
-
Aberrant homeodomain-DNA cooperative dimerization underlies distinct developmental defects in two dominant CRX retinopathy models.bioRxiv [Preprint]. 2024 Mar 14:2024.03.12.584677. doi: 10.1101/2024.03.12.584677. bioRxiv. 2024. Update in: Genome Res. 2025 Feb 14;35(2):242-256. doi: 10.1101/gr.279340.124. PMID: 38559186 Free PMC article. Updated. Preprint.
-
Aberrant homeodomain-DNA cooperative dimerization underlies distinct developmental defects in two dominant CRX retinopathy models.Genome Res. 2025 Feb 14;35(2):242-256. doi: 10.1101/gr.279340.124. Genome Res. 2025. PMID: 39715683 Free PMC article.
References
-
- Aldiri I, Xu B, Wang L, Chen X, Hiler D, Griffiths L, Valentine M, Shirinifard A, Thiagarajan S, Sablauer A, Barabas M-E, Zhang J, Johnson D, Frase S, Zhou X, Easton J, Zhang J, Mardis ER, Wilson RK, Downing JR, Dyer MA, St. Jude Children’s Research Hospital—Washington University Pediatric Cancer Genome Project The dynamic epigenetic landscape of the retina during development, reprogramming, and tumorigenesis. Neuron. 2017;94:550–568. doi: 10.1016/j.neuron.2017.04.022. - DOI - PMC - PubMed
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

