Neurodevelopmental Programming of Adiposity: Contributions to Obesity Risk
- PMID: 37971140
- PMCID: PMC10911958
- DOI: 10.1210/endrev/bnad031
Neurodevelopmental Programming of Adiposity: Contributions to Obesity Risk
Abstract
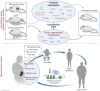
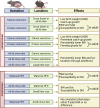
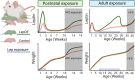
This review analyzes the published evidence regarding maternal factors that influence the developmental programming of long-term adiposity in humans and animals via the central nervous system (CNS). We describe the physiological outcomes of perinatal underfeeding and overfeeding and explore potential mechanisms that may mediate the impact of such exposures on the development of feeding circuits within the CNS-including the influences of metabolic hormones and epigenetic changes. The perinatal environment, reflective of maternal nutritional status, contributes to the programming of offspring adiposity. The in utero and early postnatal periods represent critically sensitive developmental windows during which the hormonal and metabolic milieu affects the maturation of the hypothalamus. Maternal hyperglycemia is associated with increased transfer of glucose to the fetus driving fetal hyperinsulinemia. Elevated fetal insulin causes increased adiposity and consequently higher fetal circulating leptin concentration. Mechanistic studies in animal models indicate important roles of leptin and insulin in central and peripheral programming of adiposity, and suggest that optimal concentrations of these hormones are critical during early life. Additionally, the environmental milieu during development may be conveyed to progeny through epigenetic marks and these can potentially be vertically transmitted to subsequent generations. Thus, nutritional and metabolic/endocrine signals during perinatal development can have lifelong (and possibly multigenerational) impacts on offspring body weight regulation.
Keywords: adiposity; developmental programming; epigenetics; hypothalamus; obesity.
© The Author(s) 2023. Published by Oxford University Press on behalf of the Endocrine Society.
Figures


References
-
- Fryar CD, Carroll MD, Ogden CL. Prevalence of Overweight, Obesity, and Severe Obesity among Children and Adolescents Aged 2–19 Years: United States, 1963–1965 Through 2015–2016. National Center for Health Statistics (U.S.); 2018.
-
- Must A, Must A, Jacques PF, et al. . Long-term morbidity and mortality of overweight adolescents. A follow-up of the harvard growth study of 1922 to 1935. N Engl J Med. 1992;327(19):1350‐1355. - PubMed
-
- Park MH, Falconer C, Viner RM, et al. . The impact of childhood obesity on morbidity and mortality in adulthood: a systematic review. Obes Rev. 2012;13(11):985‐1000. - PubMed
-
- Abdullah A, Wolfe R, Stoelwinder JU, et al. . The number of years lived with obesity and the risk of all-cause and cause-specific mortality. Int J Epidemiol. 2011;40(4):985‐996. - PubMed

