Distinct involvement of the cranial and spinal nerves in progressive supranuclear palsy
- PMID: 37972275
- PMCID: PMC10994524
- DOI: 10.1093/brain/awad381
Distinct involvement of the cranial and spinal nerves in progressive supranuclear palsy
Abstract
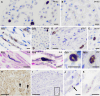
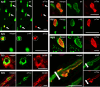
The most frequent neurodegenerative proteinopathies include diseases with deposition of misfolded tau or α-synuclein in the brain. Pathological protein aggregates in the PNS are well-recognized in α-synucleinopathies and have recently attracted attention as a diagnostic biomarker. However, there is a paucity of observations in tauopathies. To characterize the involvement of the PNS in tauopathies, we investigated tau pathology in cranial and spinal nerves (PNS-tau) in 54 tauopathy cases [progressive supranuclear palsy (PSP), n = 15; Alzheimer's disease (AD), n = 18; chronic traumatic encephalopathy (CTE), n = 5; and corticobasal degeneration (CBD), n = 6; Pick's disease, n = 9; limbic-predominant neuronal inclusion body 4-repeat tauopathy (LNT), n = 1] using immunohistochemistry, Gallyas silver staining, biochemistry, and seeding assays. Most PSP cases revealed phosphorylated and 4-repeat tau immunoreactive tau deposits in the PNS as follows: (number of tau-positive cases/available cases) cranial nerves III: 7/8 (88%); IX/X: 10/11 (91%); and XII: 6/6 (100%); anterior spinal roots: 10/10 (100%). The tau-positive inclusions in PSP often showed structures with fibrillary (neurofibrillary tangle-like) morphology in the axon that were also recognized with Gallyas silver staining. CBD cases rarely showed fine granular non-argyrophilic tau deposits. In contrast, tau pathology in the PNS was not evident in AD, CTE and Pick's disease cases. The single LNT case also showed tau pathology in the PNS. In PSP, the severity of PNS-tau involvement correlated with that of the corresponding nuclei, although, occasionally, p-tau deposits were present in the cranial nerves but not in the related brainstem nuclei. Not surprisingly, most of the PSP cases presented with eye movement disorder and bulbar symptoms, and some cases also showed lower-motor neuron signs. Using tau biosensor cells, for the first time we demonstrated seeding capacity of tau in the PNS. In conclusion, prominent PNS-tau distinguishes PSP from other tauopathies. The morphological differences of PNS-tau between PSP and CBD suggest that the tau pathology in PNS could reflect that in the central nervous system. The high frequency and early presence of tau lesions in PSP suggest that PNS-tau may have clinical and biomarker relevance.
Keywords: cranial nerve; peripheral nervous system; progressive supranuclear palsy; spinal nerve; tauopathy.
© The Author(s) 2023. Published by Oxford University Press on behalf of the Guarantors of Brain.
Conflict of interest statement
The authors report no competing interests in relation to the work described.
Figures


References
-
- Kovacs GG. Introduction: Classification of neurodegenerative diseases. In: Kovacs GG, ed. Neuropathology of neurodegenerative diseases: A practical guide. Cambridge University Press; 2014:1–7.
-
- Park A, Stacy M. Non-motor symptoms in Parkinson’s disease. J Neurol. 2009;256:293–298. - PubMed
-
- Wakabayashi K, Mori F, Tanji K, Orimo S, Takahashi H. Involvement of the peripheral nervous system in synucleinopathies, tauopathies and other neurodegenerative proteinopathies of the brain. Acta Neuropathol. 2010;120:1–12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

