Financial incentives for reduced alcohol use and increased isoniazid adherence during tuberculosis preventive therapy among people with HIV in Uganda: an open-label, factorial randomised controlled trial
- PMID: 37973340
- PMCID: PMC11005200
- DOI: 10.1016/S2214-109X(23)00436-9
Financial incentives for reduced alcohol use and increased isoniazid adherence during tuberculosis preventive therapy among people with HIV in Uganda: an open-label, factorial randomised controlled trial
Abstract
Background: Alcohol use is common among people with HIV and is a risk factor for tuberculosis disease and non-adherence to isoniazid preventive therapy (IPT). Few interventions exist to reduce alcohol use and increase IPT adherence in sub-Saharan Africa. The aim of this study was to test the hypothesis that financial incentives conditional on point-of-care negative urine alcohol biomarker testing and positive urine isoniazid testing would reduce alcohol use and increase isoniazid adherence, respectively, in people with HIV who have latent tuberculosis infection and hazardous alcohol use.
Methods: We conducted an open-label, 2×2 factorial randomised controlled trial in Uganda. Eligible for the study were non-pregnant HIV-positive adults (aged ≥18 years) prescribed antiretroviral therapy for at least 6 months, with current heavy alcohol use confirmed by urine ethyl glucuronide (biomarker of recent alcohol use) and a positive Alcohol Use Disorders Identification Test-Consumption (AUDIT-C; ≥3 for women, ≥4 for men) for the past 3 months' drinking, no history of active tuberculosis, tuberculosis treatment, or tuberculosis preventive therapy, and a positive tuberculin skin test. We randomly assigned participants (1:1:1:1) initiating 6 months of IPT to: no incentives (group 1); or incentives for recent alcohol abstinence (group 2), isoniazid adherence (group 3), or both (group 4). Escalating incentives were contingent on monthly point-of-care urine tests negative for ethyl glucuronide (groups 2 and 4), or positive on IsoScreen (biomarker of recent isoniazid use; groups 3 and 4). The primary alcohol outcome was non-hazardous use by self-report (AUDIT-C <3 for women, <4 for men) and phosphatidylethanol (PEth; past-month alcohol biomarker) <35 ng/mL at 3 months and 6 months. The primary isoniazid adherence outcome was more than 90% bottle opening of days prescribed. We performed intention-to-treat analyses. This trial is registered with ClinicalTrials.gov (NCT03492216), and is complete.
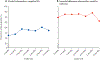
Findings: From April 16, 2018, to Aug 2, 2021, 5508 people were screened, of whom 680 were randomly assigned: 169 to group 1, 169 to group 2, 170 to group 3, and 172 to group 4. The median age of participants was 39 years (IQR 32-47), 470 (69%) were male, 598 (90%) of 663 had HIV RNA viral loads of less than 40 copies per mL, median AUDIT-C score was 6 (IQR 4-8), and median PEth was 252 ng/mL (IQR 87-579). Among 636 participants who completed the trial with alcohol use endpoint measures (group 1: 152, group 2: 159, group 3: 161, group 4: 164), non-hazardous alcohol use was more likely in the groups with incentives for alcohol abstinence (groups 2 and 4) versus no alcohol incentives (groups 1 and 3): 57 (17·6%) of 323 versus 31 (9·9%) of 313, respectively; adjusted risk difference (aRD) 7·6% (95% CI 2·7 to 12·5, p=0·0025). Among 656 participants who completed the trial with isoniazid adherence endpoint measures (group 1: 158, group 2: 163, group 3: 168, group 4: 167), incentives for isoniazid adherence did not increase adherence: 244 (72·8%) of 335 in the isoniazid incentive groups (groups 3 and 4) versus 234 (72·9%) of 321 in the no isoniazid incentive groups (groups 1 and 2); aRD -0·2% (95% CI -7·0 to 6·5, p=0·94). Overall, 53 (8%) of 680 participants discontinued isoniazid due to grade 3 or higher adverse events. There was no significant association between randomisation group and hepatotoxicity resulting in isoniazid discontinuation, after adjusting for sex and site.
Interpretation: Escalating financial incentives contingent on recent alcohol abstinence led to significantly lower biomarker-confirmed alcohol use versus control, but incentives for recent isoniazid adherence did not lead to changes in adherence. The alcohol intervention was efficacious despite less intensive frequency of incentives and clinic visits than traditional programmes for substance use, suggesting that pragmatic modifications of contingency management for resource-limited settings can have efficacy and that further evaluation of implementation is merited.
Funding: National Institute on Alcohol Abuse and Alcoholism.
Translation: For the Runyankole translation of the abstract see Supplementary Materials section.
Copyright © 2023 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY-NC-ND 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests We declare no competing interests.
Figures
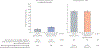
Comment in
-
Towards tuberculosis elimination: reaching the most at-risk populations with tuberculosis preventive therapy.Lancet Glob Health. 2023 Dec;11(12):e1834-e1835. doi: 10.1016/S2214-109X(23)00509-0. Lancet Glob Health. 2023. PMID: 37973327 No abstract available.
References
-
- WHO. Global status report on alcohol and health. Geneva: World Health Organization, 2018.
-
- Centers for Disease Control and Prevention. Uganda country profile https://www.cdc.gov/globalhivtb/where-we-work/uganda/uganda.html (accessed Feb 2, 2023).
-
- UNAIDS. Country: Uganda. https://www.unaids.org/en/regionscountries/countries/uganda (accessed April 4, 2023).
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

