Progranulinopathy: A diverse realm of disorders linked to progranulin imbalances
- PMID: 37981505
- PMCID: PMC10978308
- DOI: 10.1016/j.cytogfr.2023.11.001
Progranulinopathy: A diverse realm of disorders linked to progranulin imbalances
Abstract
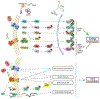
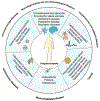
Progranulin (PGRN), encoded by the GRN gene in humans, was originally isolated as a secreted growth factor that implicates in a multitude of processes ranging from regulation of tumorigenesis, inflammation to neural proliferation. Compelling evidence indicating that GRN mutation can lead to various common neuronal degenerative diseases and rare lysosomal storage diseases. These findings have unveiled a critical role for PGRN as a lysosomal protein in maintaining lysosomal function. The phenotypic spectrum of PGRN imbalance has expanded to encompass a broad spectrum of diseases, including autoimmune diseases, metabolic, musculoskeletal and cardiovascular diseases. These diseases collectively referred to as Progranulinopathy- a term encompasses the wide spectrum of disorders influenced by PGRN imbalance. Unlike its known extracellular function as a growth factor-like molecule associated with multiple membrane receptors, PGRN also serves as an intracellular co-chaperone engaged in the folding and traffic of its associated proteins, particularly the lysosomal hydrolases. This chaperone activity is required for PGRN to exert its diverse functions across a broad range of diseases, encompassing both the central nervous system and peripheral systems. In this comprehensive review, we present an update of the emerging role of PGRN in Progranulinopathy, with special focus on elucidating the intricate interplay between PGRN and a diverse array of proteins at various levels, ranging from extracellular fluids and intracellular components, as well as various pathophysiological processes involved. This review seeks to offer a comprehensive grasp of PGRN's diverse functions, aiming to unveil intricate mechanisms behind Progranulinopathy and open doors for future research endeavors.
Keywords: Autoimmune diseases; Cancer; Lysosomal Storage Diseases; Neurodegenerative Diseases; Progranulin; Progranulinopathy.
Copyright © 2023 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare no competing interests.
Figures
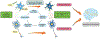


Similar articles
-
Progranulin: A conductor of receptors orchestra, a chaperone of lysosomal enzymes and a therapeutic target for multiple diseases.Cytokine Growth Factor Rev. 2019 Feb;45:53-64. doi: 10.1016/j.cytogfr.2019.01.002. Epub 2019 Jan 30. Cytokine Growth Factor Rev. 2019. PMID: 30733059 Free PMC article. Review.
-
Early lysosomal maturation deficits in microglia triggers enhanced lysosomal activity in other brain cells of progranulin knockout mice.Mol Neurodegener. 2018 Sep 4;13(1):48. doi: 10.1186/s13024-018-0281-5. Mol Neurodegener. 2018. PMID: 30180904 Free PMC article.
-
The lysosomal function of progranulin, a guardian against neurodegeneration.Acta Neuropathol. 2018 Jul;136(1):1-17. doi: 10.1007/s00401-018-1861-8. Epub 2018 May 9. Acta Neuropathol. 2018. PMID: 29744576 Free PMC article. Review.
-
Progranulin acts as a shared chaperone and regulates multiple lysosomal enzymes.Genes Dis. 2017 Sep;4(3):125-126. doi: 10.1016/j.gendis.2017.05.001. Epub 2017 Jun 23. Genes Dis. 2017. PMID: 28944282 Free PMC article.
-
Progranulin regulates neuronal outgrowth independent of sortilin.Mol Neurodegener. 2012 Jul 10;7:33. doi: 10.1186/1750-1326-7-33. Mol Neurodegener. 2012. PMID: 22781549 Free PMC article.
Cited by
-
The antioxidant stress effect of granulin precursor in vitiligo.Sci Rep. 2025 May 25;15(1):18189. doi: 10.1038/s41598-025-03486-7. Sci Rep. 2025. PMID: 40415096 Free PMC article.
-
Ion channels in osteoarthritis: emerging roles and potential targets.Nat Rev Rheumatol. 2024 Sep;20(9):545-564. doi: 10.1038/s41584-024-01146-0. Epub 2024 Aug 9. Nat Rev Rheumatol. 2024. PMID: 39122910 Free PMC article. Review.
-
Progranulin deficiency in the brain: the interplay between neuronal and non-neuronal cells.Transl Neurodegener. 2025 Apr 16;14(1):18. doi: 10.1186/s40035-025-00475-8. Transl Neurodegener. 2025. PMID: 40234992 Free PMC article. Review.
-
Versatility of 14-3-3 proteins and their roles in bone and joint-related diseases.Bone Res. 2024 Oct 15;12(1):58. doi: 10.1038/s41413-024-00370-4. Bone Res. 2024. PMID: 39406741 Free PMC article. Review.
-
Consistent efficacy outcomes between phase 2 and phase 3 trials in Crohn's disease or ulcerative colitis in adults: a meta-analysis.Inflamm Res. 2024 Jun;73(6):915-928. doi: 10.1007/s00011-024-01874-9. Epub 2024 Apr 8. Inflamm Res. 2024. PMID: 38587530
References
-
- Zanocco-Marani T, Bateman A, Romano G, Valentinis B, He ZH, Baserga R, Biological activities and signaling pathways of the granulin/epithelin precursor, Cancer Research 59(20) (1999) 5331–5340. - PubMed
-
- Hrabal R, Chen Z, James S, Bennett HP, Ni F, The hairpin stack fold, a novel protein architecture for a new family of protein growth factors, Nat Struct Biol 3(9) (1996) 747–752. - PubMed
-
- He Z, Bateman A, Progranulin gene expression regulates epithelial cell growth and promotes tumor growth in vivo, Cancer Research 59(13) (1999) 3222–3229. - PubMed
-
- Baker M, Mackenzie IR, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C, Snowden J, Adamson J, Sadovnick AD, Rollinson S, Cannon A, Dwosh E, Neary D, Melquist S, Richardson A, Dickson D, Berger Z, Eriksen J, Robinson T, Zehr C, Dickey CA, Crook R, McGowan E, Mann D, Boeve B, Feldman H, Hutton M, Mutations in progranulin cause tau-negative frontotemporal dementia linked to chromosome 17, Nature 442(7105) (2006) 916–919. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

