Synthesis and biological evaluation of structurally diverse 6-aryl-3-aroyl-indole analogues as inhibitors of tubulin polymerization
- PMID: 37984295
- PMCID: PMC11019941
- DOI: 10.1016/j.ejmech.2023.115794
Synthesis and biological evaluation of structurally diverse 6-aryl-3-aroyl-indole analogues as inhibitors of tubulin polymerization
Abstract
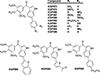
The synthesis and evaluation of small-molecule inhibitors of tubulin polymerization remains a promising approach for the development of new therapeutic agents for cancer treatment. The natural products colchicine and combretastatin A-4 (CA4) inspired significant drug discovery campaigns targeting the colchicine site located on the beta-subunit of the tubulin heterodimer, but so far these efforts have not yielded an approved drug for cancer treatment in human patients. Interest in the colchicine site was enhanced by the discovery that a subset of colchicine site agents demonstrated dual functionality as both potent antiproliferative agents and effective vascular disrupting agents (VDAs). Our previous studies led to the discovery and development of a 2-aryl-3-aroyl-indole analogue (OXi8006) that inhibited tubulin polymerization and demonstrated low nM IC50 values against a variety of human cancer cell lines. A water-soluble phosphate prodrug salt (OXi8007), synthesized from OXi8006, displayed promising vascular disrupting activity in mouse models of cancer. To further extend structure-activity relationship correlations, a series of 6-aryl-3-aroyl-indole analogues was synthesized and evaluated for their inhibition of tubulin polymerization and cytotoxicity against human cancer cell lines. Several structurally diverse molecules in this small library were strong inhibitors of tubulin polymerization and of MCF-7 and MDA-MB-231 human breast cancer cells. One of the most promising analogues (KGP591) caused significant G2/M arrest of MDA-MB-231 cells, disrupted microtubule structure and cell morphology in MDA-MB-231 cells, and demonstrated significant inhibition of MDA-MB-231 cell migration in a wound healing (scratch) assay. A phosphate prodrug salt, KGP618, synthesized from its parent phenolic precursor, KGP591, demonstrated significant reduction in bioluminescence signal when evaluated in vivo against an orthotopic model of kidney cancer (RENCA-luc) in BALB/c mice, indicative of VDA efficacy. The most active compounds from this series offer promise as anticancer therapeutic agents.
Keywords: Antiproliferative agents; Indole synthesis; Inhibitors of cell migration; Inhibitors of tubulin polymerization; Molecular docking; Vascular disrupting agents.
Copyright © 2023 Elsevier Masson SAS. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





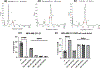


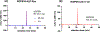




Similar articles
-
Design, Synthesis and Biological Evaluation of 2-Phenyl Indole Analogues of OXi8006 as Colchicine Site Inhibitors of Tubulin Polymerization and Vascular Disrupting Agents.Bioorg Med Chem. 2025 Feb 1;118:117981. doi: 10.1016/j.bmc.2024.117981. Epub 2024 Nov 7. Bioorg Med Chem. 2025. PMID: 39667146
-
Structure Guided Design, Synthesis, and Biological Evaluation of Oxetane-Containing Indole Analogues.Bioorg Med Chem. 2023 Sep 7;92:117400. doi: 10.1016/j.bmc.2023.117400. Epub 2023 Jun 29. Bioorg Med Chem. 2023. PMID: 37556912 Free PMC article.
-
Synthesis and Preclinical Evaluation of Indole Triazole Conjugates as Microtubule Targeting Agents that are Effective against MCF-7 Breast Cancer Cell Lines.Anticancer Agents Med Chem. 2021;21(8):1047-1055. doi: 10.2174/1871520620666200925102940. Anticancer Agents Med Chem. 2021. PMID: 32981511
-
Synthesis of a 2-aryl-3-aroyl indole salt (OXi8007) resembling combretastatin A-4 with application as a vascular disrupting agent.J Nat Prod. 2013 Sep 27;76(9):1668-78. doi: 10.1021/np400374w. Epub 2013 Sep 9. J Nat Prod. 2013. PMID: 24016002 Free PMC article.
-
Discovery of novel thienopyridine indole derivatives as inhibitors of tubulin polymerization targeting the colchicine-binding site with potent anticancer activities.Eur J Med Chem. 2025 Mar 15;286:117314. doi: 10.1016/j.ejmech.2025.117314. Epub 2025 Jan 23. Eur J Med Chem. 2025. PMID: 39874633
Cited by
-
Benzosuberene and Tetracyclic Analogues as Colchicine Site Inhibitors of Tubulin Polymerization.ACS Med Chem Lett. 2025 May 26;16(6):1098-1107. doi: 10.1021/acsmedchemlett.5c00129. eCollection 2025 Jun 12. ACS Med Chem Lett. 2025. PMID: 40529069
-
Evaluating Therapeutic Efficacy of the Vascular Disrupting Agent OXi8007 Against Kidney Cancer in Mice.Cancers (Basel). 2025 Feb 24;17(5):771. doi: 10.3390/cancers17050771. Cancers (Basel). 2025. PMID: 40075618 Free PMC article.
-
Indole Compounds in Oncology: Therapeutic Potential and Mechanistic Insights.Pharmaceuticals (Basel). 2024 Jul 10;17(7):922. doi: 10.3390/ph17070922. Pharmaceuticals (Basel). 2024. PMID: 39065774 Free PMC article. Review.
-
Indole Derivatives: A Versatile Scaffold in Modern Drug Discovery-An Updated Review on Their Multifaceted Therapeutic Applications (2020-2024).Molecules. 2024 Oct 9;29(19):4770. doi: 10.3390/molecules29194770. Molecules. 2024. PMID: 39407697 Free PMC article. Review.
-
VERU-111, an orally available tubulin inhibitor, suppresses ovarian tumor growth and metastasis.J Pharmacol Exp Ther. 2025 Jan;392(1):100006. doi: 10.1124/jpet.124.002298. Epub 2024 Nov 22. J Pharmacol Exp Ther. 2025. PMID: 39893008 Free PMC article.
References
-
- Alpizar-Pedraza D, Veulens AD, Araujo EC, Piloto-Ferrer J, Sanchez-Lamar A, Microtubules destabilizing agents binding sites in tubulin, J Mol Struct, 1259 (2022) 132723.
-
- Hastie SB, Interactions of colchicine with tubulin, Pharmacol Ther, 51 (1991) 377–401. - PubMed
-
- Pettit GR, Lippert JW 3rd, Antineoplastic agents 429. Syntheses of the combretastatin A-1 and combretastatin B-1 prodrugs, Anticancer Drug Des, 15 (2000) 203–216. - PubMed
-
- Pettit G, Singh S, Hamel E, Lin C, Alberts D, Garcia-Kendal D, Antitumor activity of combretastatin-A4 phosphate, a natural product tubulin inhibitor, Experientia, 45 (1989) 209211.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

