Managing oral biofilms to avoid enamel demineralization during fixed orthodontic treatment
- PMID: 37989574
- PMCID: PMC10663579
- DOI: 10.4041/kjod23.184
Managing oral biofilms to avoid enamel demineralization during fixed orthodontic treatment
Abstract
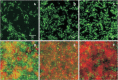
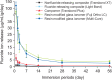
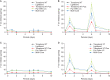
Enamel demineralization represents the most prevalent complication arising from fixed orthodontic treatment. Its main etiology is the development of cariogenic biofilms formed around orthodontic appliances. Ordinarily, oral biofilms exist in a dynamic equilibrium with the host's defense mechanisms. However, the equilibrium can be disrupted by environmental changes, such as the introduction of a fixed orthodontic appliance, resulting in a shift in the biofilm's microbial composition from non-pathogenic to pathogenic. This alteration leads to an increased prevalence of cariogenic bacteria, notably mutans streptococci, within the biofilm. This article examines the relationships between oral biofilms and orthodontic appliances, with a particular focus on strategies for effectively managing oral biofilms to mitigate enamel demineralization around orthodontic appliances.
Keywords: Biofilm; Biomaterial science; Enamel demineralization; Mutans streptococci.
Conflict of interest statement
No potential conflict of interest relevant to this article was reported.
Figures





References
-
- Mizrahi E. Enamel demineralization following orthodontic treatment. Am J Orthod. 1982;82:62–7. doi: 10.1016/0002-9416(82)90548-6. https://doi.org/10.1016/0002-9416(82)90548-6. - DOI - PubMed
-
- Lucchese A, Gherlone E. Prevalence of white-spot lesions before and during orthodontic treatment with fixed appliances. Eur J Orthod. 2013;35:664–8. doi: 10.1093/ejo/cjs070. https://doi.org/10.1093/ejo/cjs070. - DOI - PubMed
-
- Sharab L, Loss C, Jensen D, Kluemper GT, Alotaibi M, Nagaoka H. Prevalence of white spot lesions and gingival index during orthodontic treatment in an academic setting. Am J Orthod Dentofacial Orthop. 2023;163:835–42. doi: 10.1016/j.ajodo.2022.08.023. https://doi.org/10.1016/j.ajodo.2022.08.023. - DOI - PubMed
-
- Sonesson M, Bergstrand F, Gizani S, Twetman S. Management of post-orthodontic white spot lesions: an updated systematic review. Eur J Orthod. 2017;39:116–21. doi: 10.1093/ejo/cjw023. https://doi.org/10.1093/ejo/cjw023. - DOI - PubMed
-
- Ulukapi H, Koray F, Efes B. Monitoring the caries risk of orthodontic patients. Quintessence Int. 1997;28:27–9. https://pubmed.ncbi.nlm.nih.gov/10332351/ - PubMed
Publication types
LinkOut - more resources
Full Text Sources
