Spray-dried nanocrystal-loaded polymer microparticles for long-term release local therapies: an opportunity for poorly soluble drugs
- PMID: 37994039
- PMCID: PMC10987046
- DOI: 10.1080/10717544.2023.2284683
Spray-dried nanocrystal-loaded polymer microparticles for long-term release local therapies: an opportunity for poorly soluble drugs
Abstract
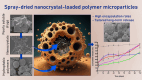
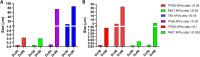
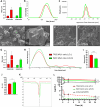
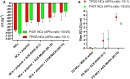
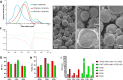
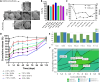
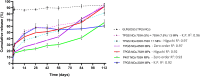
Nano- and micro-technologies can salvage drugs with very low solubility that were doomed to pre-clinical and clinical failure. A unique design approach to develop drug nanocrystals (NCs) loaded in extended release polymeric microparticles (MPs) for local treatments is presented here through the case of a potential osteoarthritis (OA) drug candidate for intra-articular (IA) administration. Optimizing a low-shear wet milling process allowed the production of NCs that can be subsequently freeze-dried (FD) and redispersed in a hydrophobic polymer-organic solvent solution to form spray-dried MPs. Results demonstrated a successful development of a ready-to-upscale formulation containing PLGA MPs with high drug NC encapsulation rates that showed a continuous and controlled drug release profile over four months. The screenings and procedures described allowed for identifying and overcoming common difficulties and challenges raised along the drug reduction to nano-size and spray-drying process. Above all, the technical knowledge acquired is intended for formulation scientists aiming to improve the therapeutic perspectives of poorly soluble drugs.
Keywords: Osteoarthritis; PLGA; microparticles; nanocrystals; spray-drying.
Conflict of interest statement
No potential conflict of interest was reported by the authors. Gaby Thonus owns subscription rights of Galapagos. Sam Corveleyn and Joke Meeus are employee of Galapagos and own subscription rights in the company.
Figures

Similar articles
-
Enhanced encapsulation and bioavailability of breviscapine in PLGA microparticles by nanocrystal and water-soluble polymer template techniques.Eur J Pharm Biopharm. 2017 Jun;115:177-185. doi: 10.1016/j.ejpb.2017.02.021. Epub 2017 Mar 2. Eur J Pharm Biopharm. 2017. PMID: 28263795
-
Insight into Nanoscale Network of Spray-Dried Polymeric Particles: Role of Polymer Molecular Conformation.ACS Appl Mater Interfaces. 2018 Oct 31;10(43):36686-36692. doi: 10.1021/acsami.8b12475. Epub 2018 Oct 22. ACS Appl Mater Interfaces. 2018. PMID: 30211530
-
In vivo evaluation of solid lipid microparticles and hybrid polymer-lipid microparticles for sustained delivery of leuprolide.Eur J Pharm Biopharm. 2019 Sep;142:315-321. doi: 10.1016/j.ejpb.2019.07.010. Epub 2019 Jul 9. Eur J Pharm Biopharm. 2019. PMID: 31299277
-
Nano Spray-Dried Drugs for Oral Administration: A Review.Assay Drug Dev Technol. 2021 Oct;19(7):412-441. doi: 10.1089/adt.2021.053. Epub 2021 Sep 21. Assay Drug Dev Technol. 2021. PMID: 34550790 Review.
-
Nano/micro technologies for delivering macromolecular therapeutics using poly(D,L-lactide-co-glycolide) and its derivatives.J Control Release. 2008 Feb 11;125(3):193-209. doi: 10.1016/j.jconrel.2007.09.013. Epub 2007 Oct 22. J Control Release. 2008. PMID: 18083265 Review.
Cited by
-
Recent advances in nanocrystals for arthritis drug delivery.Expert Opin Drug Deliv. 2025 Jul;22(7):1031-1042. doi: 10.1080/17425247.2025.2505758. Epub 2025 May 31. Expert Opin Drug Deliv. 2025. PMID: 40357685 Free PMC article. Review.
-
Nanocrystals and nanosuspensions: an exploration from classic formulations to advanced drug delivery systems.Drug Deliv Transl Res. 2024 Dec;14(12):3438-3451. doi: 10.1007/s13346-024-01559-0. Epub 2024 Mar 7. Drug Deliv Transl Res. 2024. PMID: 38451440 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
