Talniflumate abrogates mucin immune suppressive barrier improving efficacy of gemcitabine and nab-paclitaxel treatment in pancreatic cancer
- PMID: 37996891
- PMCID: PMC10668479
- DOI: 10.1186/s12967-023-04733-z
Talniflumate abrogates mucin immune suppressive barrier improving efficacy of gemcitabine and nab-paclitaxel treatment in pancreatic cancer
Abstract
Background: Pancreatic ductal adenocarcinoma (PDAC) is a lethal disease. This is due to its aggressive course, late diagnosis and its intrinsic drugs resistance. The complexity of the tumor, in terms of cell components and heterogeneity, has led to the approval of few therapies with limited efficacy. The study of the early stages of carcinogenesis provides the opportunity for the identification of actionable pathways that underpin therapeutic resistance.
Methods: We analyzed 43 Intraductal papillary mucinous neoplasms (IPMN) (12 Low-grade and 31 High-grade) by Spatial Transcriptomics. Mouse and human pancreatic cancer organoids and T cells interaction platforms were established to test the role of mucins expression on T cells activity. Syngeneic mouse model of PDAC was used to explore the impact of mucins downregulation on standard therapy efficacy.
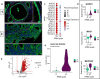
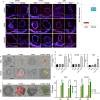
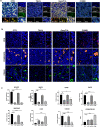
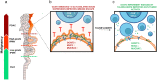
Results: Spatial transcriptomics showed that mucin O-glycosylation pathway is increased in the progression from low-grade to high-grade IPMN. We identified GCNT3, a master regulator of mucins expression, as an actionable target of this pathway by talniflumate. We showed that talniflumate impaired mucins expression increasing T cell activation and recognition using both mouse and human organoid interaction platforms. In vivo experiments showed that talniflumate was able to increase the efficacy of the chemotherapy by boosting immune infiltration.
Conclusions: Finally, we demonstrated that combination of talniflumate, an anti-inflammatory drug, with chemotherapy effectively improves anti-tumor effect in PDAC.
Keywords: Intraductal mucinous neoplasms (IPMNs); Organoid interaction platform; Pancreatic ductal adenocarcinoma (PDAC); Spatial transcriptomics; Syngeneic mouse models.
© 2023. The Author(s).
Conflict of interest statement
The authors have no competing interests to disclose.
Figures



References
-
- Chun JW, Lee SH, Kim JS, Park N, Huh G, Cho IR, et al. Comparison between FOLFIRINOX and gemcitabine plus nab-paclitaxel including sequential treatment for metastatic pancreatic cancer: a propensity score matching approach. BMC Cancer. 2021;21:537. doi: 10.1186/s12885-021-08277-7. - DOI - PMC - PubMed
-
- Beatty PL, van der Geest R, Hashash JG, Kimura T, Gutkin D, Brand RE, et al. Immunobiology and immunosurveillance in patients with intraductal papillary mucinous neoplasms (IPMNs), premalignant precursors of pancreatic adenocarcinomas. Cancer Immunol Immunother. 2016;65:771–778. doi: 10.1007/s00262-016-1838-1. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 23681/Associazione Italiana per la Ricerca sul Cancro
- 26330/Associazione Italiana per la Ricerca sul Cancro
- 20583/Associazione Italiana per la Ricerca sul Cancro
- 24519/Associazione Italiana per la Ricerca sul Cancro
- J38D19000690001/Agenzia Italiana del Farmaco, Ministero della Salute
- 2019-12369662/Agenzia Italiana del Farmaco, Ministero della Salute
- PRIN 2022 Prot. 2022P79F9N/Ministero dell'Università e della Ricerca
- PRIN 2022 PNRR Prot. P2022LN3KS/Ministero dell'Università e della Ricerca
- B39J22001200001/Ministero dell'Università e della Ricerca
- B33C22000630001/Ministero dell'Università e della Ricerca
- B38D19000140006/Ministero dell'Istruzione, dell'Università e della Ricerca
LinkOut - more resources
Full Text Sources
Medical

