Krüppel-like Factor 5 Plays an Important Role in the Pathogenesis of Chronic Pancreatitis
- PMID: 38001687
- PMCID: PMC10670257
- DOI: 10.3390/cancers15225427
Krüppel-like Factor 5 Plays an Important Role in the Pathogenesis of Chronic Pancreatitis
Abstract
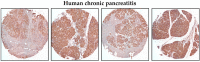
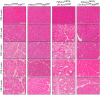
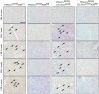
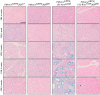
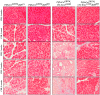
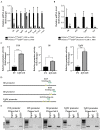
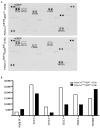
Chronic pancreatitis results in the formation of pancreatic intraepithelial neoplasia (PanIN) and poses a risk of developing pancreatic cancer. Our previous study demonstrated that Krüppel-like factor 5 (KLF5) is necessary for forming acinar-to-ductal metaplasia (ADM) in acute pancreatitis. Here, we investigated the role of KLF5 in response to chronic injury in the pancreas. Human tissues originating from chronic pancreatitis patients showed increased levels of epithelial KLF5. An inducible genetic model combining the deletion of Klf5 and the activation of KrasG12D mutant expression in pancreatic acinar cells together with chemically induced chronic pancreatitis was used. The chronic injury resulted in increased levels of KLF5 in both control and KrasG12D mutant mice. Furthermore, it led to numerous ADM and PanIN lesions and extensive fibrosis in the KRAS mutant mice. In contrast, pancreata with Klf5 loss (with or without KrasG12D) failed to develop ADM, PanIN, or significant fibrosis. Furthermore, the deletion of Klf5 reduced the expression level of cytokines and fibrotic components such as Il1b, Il6, Tnf, Tgfb1, Timp1, and Mmp9. Notably, using ChIP-PCR, we showed that KLF5 binds directly to the promoters of Il1b, Il6, and Tgfb1 genes. In summary, the inactivation of Klf5 inhibits ADM and PanIN formation and the development of pancreatic fibrosis.
Keywords: Kirsten rat sarcoma viral oncogene homolog; Krüppel-like factor 5; acinar-to-ductal metaplasia; pancreatic intraepithelial neoplasia.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Krüppel-like Factor 5, Increased in Pancreatic Ductal Adenocarcinoma, Promotes Proliferation, Acinar-to-Ductal Metaplasia, Pancreatic Intraepithelial Neoplasia, and Tumor Growth in Mice.Gastroenterology. 2018 Apr;154(5):1494-1508.e13. doi: 10.1053/j.gastro.2017.12.005. Epub 2017 Dec 15. Gastroenterology. 2018. PMID: 29248441 Free PMC article.
-
ANGPTL4 accelerates KRASG12D-Induced acinar to ductal metaplasia and pancreatic carcinogenesis.Cancer Lett. 2021 Oct 28;519:185-198. doi: 10.1016/j.canlet.2021.07.036. Epub 2021 Jul 24. Cancer Lett. 2021. PMID: 34311032
-
Hes1 plays an essential role in Kras-driven pancreatic tumorigenesis.Oncogene. 2019 May;38(22):4283-4296. doi: 10.1038/s41388-019-0718-5. Epub 2019 Jan 31. Oncogene. 2019. PMID: 30705405
-
Therapeutic potential of targeting acinar cell reprogramming in pancreatic cancer.World J Gastroenterol. 2016 Aug 21;22(31):7046-57. doi: 10.3748/wjg.v22.i31.7046. World J Gastroenterol. 2016. PMID: 27610015 Free PMC article. Review.
-
Macrophage-induced reactive oxygen species in the initiation of pancreatic cancer: a mini-review.Front Immunol. 2024 Mar 21;15:1278807. doi: 10.3389/fimmu.2024.1278807. eCollection 2024. Front Immunol. 2024. PMID: 38576613 Free PMC article. Review.
Cited by
-
Activin A signaling stimulates neutrophil activation and macrophage migration in pancreatitis.Sci Rep. 2024 Apr 23;14(1):9382. doi: 10.1038/s41598-024-60065-y. Sci Rep. 2024. PMID: 38654064 Free PMC article.
References
-
- Peery A.F., Crockett S.D., Barritt A.S., Dellon E.S., Eluri S., Gangarosa L.M., Jensen E.T., Lund J.L., Pasricha S., Runge T., et al. Burden of Gastrointestinal, Liver, and Pancreatic Diseases in the United States. Gastroenterology. 2015;149:1731–1741.e3. doi: 10.1053/j.gastro.2015.08.045. - DOI - PMC - PubMed
-
- Gapp J., Chandra S. Acute Pancreatitis. StatPearls; Treasure Island, FL, USA: 2022.
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

