C-Reactive Protein as an Early Predictor of Efficacy in Advanced Non-Small-Cell Lung Cancer Patients: A Tumor Dynamics-Biomarker Modeling Framework
- PMID: 38001689
- PMCID: PMC10670607
- DOI: 10.3390/cancers15225429
C-Reactive Protein as an Early Predictor of Efficacy in Advanced Non-Small-Cell Lung Cancer Patients: A Tumor Dynamics-Biomarker Modeling Framework
Abstract
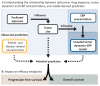
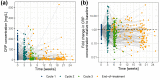
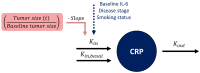
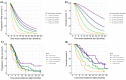
In oncology, longitudinal biomarkers reflecting the patient's status and disease evolution can offer reliable predictions of the patient's response to treatment and prognosis. By leveraging clinical data in patients with advanced non-small-cell lung cancer receiving first-line chemotherapy, we aimed to develop a framework combining anticancer drug exposure, tumor dynamics (RECIST criteria), and C-reactive protein (CRP) concentrations, using nonlinear mixed-effects models, to evaluate and quantify by means of parametric time-to-event models the significance of early longitudinal predictors of progression-free survival (PFS) and overall survival (OS). Tumor dynamics was characterized by a tumor size (TS) model accounting for anticancer drug exposure and development of drug resistance. CRP concentrations over time were characterized by a turnover model. An x-fold change in TS from baseline linearly affected CRP production. CRP concentration at treatment cycle 3 (day 42) and the difference between CRP concentration at treatment cycles 3 and 2 were the strongest predictors of PFS and OS. Measuring longitudinal CRP allows for the monitoring of inflammatory levels and, along with its reduction across treatment cycles, presents a promising prognostic marker. This framework could be applied to other treatment modalities such as immunotherapies or targeted therapies allowing the timely identification of patients at risk of early progression and/or short survival to spare them unnecessary toxicities and provide alternative treatment decisions.
Keywords: C-reactive protein; biomarkers; non-small-cell lung cancer; nonlinear mixed-effects modeling; overall survival; prognosis; progression-free survival; time-to-event analysis.
Conflict of interest statement
C.K. and W.H. report grants from an industry consortium (AbbVie Deutschland GmbH & Co. K.G., Astra Zeneca, Boehringer Ingelheim Pharma GmbH & Co. K.G., F. Hoffmann-La Roche Ltd., Merck KGaA, Novo Nordisk, and Sanofi) for the PharMetrX program. C.K. reports grants from the Innovative Medicines Initiative-Joint Undertaking (“DDMore”), from H2020-EU.3.1.3 (“FAIR”), Diurnal Ltd. and the Federal Ministry of Education and Research within the Joint Programming Initiative on Antimicrobial Resistance Initiative (“JPIAMR”), all outside the submitted work. M.J. reports advisory roles (institutional): Novartis, Astra Zeneca, Basilea Pharmaceutica, Bayer, BMS, Debiopharm, MSD, Roche, Sanofi; research funding: Swiss Cancer Research; travel grants: Roche, Sanofi, Takeda. A.P.-P. is an employee of Boehringer Ingelheim Pharma GmbH & Co. KG. S.H. has received research funding or honoraria from Roche Diagnostics, Sysmex, Thermo, VolitionRx, Mikrogen, Trillium, Medica, and Instand and is founder of SFZ BioCoDE and CEBIO. All other authors declare that they have no conflicts of interest that might be relevant to the contents of this manuscript.
Figures


Similar articles
-
C-Reactive Protein (CRP) Levels in Immune Checkpoint Inhibitor Response and Progression in Advanced Non-Small Cell Lung Cancer: A Bi-Center Study.Cancers (Basel). 2020 Aug 17;12(8):2319. doi: 10.3390/cancers12082319. Cancers (Basel). 2020. PMID: 32824580 Free PMC article.
-
C-reactive protein at 1 month after treatment of nivolumab as a predictive marker of efficacy in advanced renal cell carcinoma.Cancer Chemother Pharmacol. 2020 Jul;86(1):75-85. doi: 10.1007/s00280-020-04088-y. Epub 2020 Jun 14. Cancer Chemother Pharmacol. 2020. PMID: 32537714
-
Association of Survival and Immune-Related Biomarkers With Immunotherapy in Patients With Non-Small Cell Lung Cancer: A Meta-analysis and Individual Patient-Level Analysis.JAMA Netw Open. 2019 Jul 3;2(7):e196879. doi: 10.1001/jamanetworkopen.2019.6879. JAMA Netw Open. 2019. PMID: 31290993 Free PMC article.
-
Overview of resistance to systemic therapy in patients with breast cancer.Adv Exp Med Biol. 2007;608:1-22. doi: 10.1007/978-0-387-74039-3_1. Adv Exp Med Biol. 2007. PMID: 17993229 Review.
-
Support to early clinical decisions in drug development and personalised medicine with checkpoint inhibitors using dynamic biomarker-overall survival models.Br J Cancer. 2023 Oct;129(9):1383-1388. doi: 10.1038/s41416-023-02190-5. Epub 2023 Feb 10. Br J Cancer. 2023. PMID: 36765177 Free PMC article. Review.
Cited by
-
Population Pharmacokinetics and Exposure-Response of Subcutaneous Atezolizumab in Patients With Non-Small Cell Lung Cancer.CPT Pharmacometrics Syst Pharmacol. 2025 Apr;14(4):726-737. doi: 10.1002/psp4.13310. Epub 2025 Feb 5. CPT Pharmacometrics Syst Pharmacol. 2025. PMID: 39906948 Free PMC article. Clinical Trial.
-
A Multiple Imputation Workflow for Handling Missing Covariate Data in Pharmacometrics Modeling.CPT Pharmacometrics Syst Pharmacol. 2025 Jun;14(6):991-1005. doi: 10.1002/psp4.70039. Epub 2025 May 29. CPT Pharmacometrics Syst Pharmacol. 2025. PMID: 40439087 Free PMC article.
-
C-reactive protein as robust laboratory value associated with prognosis in patients with stage III non-small cell lung cancer (NSCLC) treated with definitive radiochemotherapy.Sci Rep. 2024 Jun 14;14(1):13765. doi: 10.1038/s41598-024-64302-2. Sci Rep. 2024. PMID: 38877146 Free PMC article.
-
The Prognostic Value of B12/CRP Ratio (BCR) in Patients Diagnosed With Solid Cancer.Clin Med Insights Oncol. 2025 Aug 10;19:11795549251360891. doi: 10.1177/11795549251360891. eCollection 2025. Clin Med Insights Oncol. 2025. PMID: 40799500 Free PMC article.
-
The Significance of C-Reactive Protein Value and Tumor Grading for Malignant Tumors: A Systematic Review.Diagnostics (Basel). 2024 Sep 19;14(18):2073. doi: 10.3390/diagnostics14182073. Diagnostics (Basel). 2024. PMID: 39335753 Free PMC article. Review.
References
-
- American Lung Association . State of Lung Cancer. American Lung Association; Chicago, IL, USA: 2022. [(accessed on 29 August 2023)]. Available online: https://www.lung.org/getmedia/647c433b-4cbc-4be6-9312-2fa9a449d489/solc-....
-
- Van Laar M., van Amsterdam W.A.C., van Lindert A.S.R., de Jong P.A., Verhoeff J.J.C. Prognostic Factors for Overall Survival of Stage III Non-Small Cell Lung Cancer Patients on Computed Tomography: A Systematic Review and Meta-Analysis. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 2020;151:152–175. doi: 10.1016/j.radonc.2020.07.030. - DOI - PubMed
-
- Katsurada M., Nagano T., Tachihara M., Kiriu T., Furukawa K., Koyama K., Otoshi T., Sekiya R., Hazama D., Tamura D. Baseline Tumor Size as a Predictive and Prognostic Factor of Immune Checkpoint Inhibitor Therapy for Non-Small Cell Lung Cancer. Anticancer Res. 2019;39:815–825. doi: 10.21873/anticanres.13180. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

