Regucalcin Is a Potential Regulator in Human Cancer: Aiming to Expand into Cancer Therapy
- PMID: 38001749
- PMCID: PMC10670417
- DOI: 10.3390/cancers15225489
Regucalcin Is a Potential Regulator in Human Cancer: Aiming to Expand into Cancer Therapy
Abstract
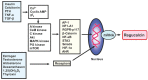
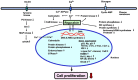
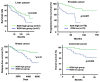
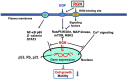
Regucalcin, a calcium-binding protein lacking the EF-hand motif, was initially discovered in 1978. Its name is indicative of its function in calcium signaling regulation. The rgn gene encodes for regucalcin and is situated on the X chromosome in both humans and vertebrates. Regucalcin regulates pivotal enzymes involved in signal transduction and has an inhibitory function, which includes protein kinases, protein phosphatases, cysteinyl protease, nitric oxide dynthetase, aminoacyl-transfer ribonucleic acid (tRNA) synthetase, and protein synthesis. This cytoplasmic protein is transported to the nucleus where it regulates deoxyribonucleic acid and RNA synthesis as well as gene expression. Overexpression of regucalcin inhibits proliferation in both normal and cancer cells in vitro, independent of apoptosis. During liver regeneration in vivo, endogenous regucalcin suppresses cell growth when overexpressed. Regucalcin mRNA and protein expressions are significantly downregulated in tumor tissues of patients with various types of cancers. Patients exhibiting upregulated regucalcin in tumor tissue have shown prolonged survival. The decrease of regucalcin expression is linked to the advancement of cancer. Overexpression of regucalcin carries the potential for preventing and treating carcinogenesis. Additionally, extracellular regucalcin has displayed control over various types of human cancer cells. Regucalcin may hold a prominent role as a regulatory factor in cancer development. Supplying the regucalcin gene could prove to be a valuable asset in cancer treatment. The therapeutic value of regucalcin suggests its potential significance in treating cancer patients. This review delves into the most recent research on the regulatory role of regucalcin in human cancer development, providing a novel approach for treatment.
Keywords: cancer suppressor; carcinogenesis; cell proliferation; cell signaling; gene therapy; regucalcin.
Conflict of interest statement
The author declares no conflict of interest.
Figures

Similar articles
-
Role of regucalcin in cell nuclear regulation: involvement as a transcription factor.Cell Tissue Res. 2013 Nov;354(2):331-41. doi: 10.1007/s00441-013-1665-z. Epub 2013 Jun 22. Cell Tissue Res. 2013. PMID: 23793546 Review.
-
The anti-apoptotic effect of regucalcin is mediated through multisignaling pathways.Apoptosis. 2013 Oct;18(10):1145-53. doi: 10.1007/s10495-013-0859-x. Apoptosis. 2013. PMID: 23670020 Free PMC article. Review.
-
Suppressive role of regucalcin in liver cell proliferation: involvement in carcinogenesis.Cell Prolif. 2013 Jun;46(3):243-53. doi: 10.1111/cpr.12036. Cell Prolif. 2013. PMID: 23692083 Free PMC article. Review.
-
Involvement of regucalcin in lipid metabolism and diabetes.Metabolism. 2013 Aug;62(8):1045-51. doi: 10.1016/j.metabol.2013.01.023. Epub 2013 Feb 28. Metabolism. 2013. PMID: 23453039 Review.
-
Regucalcin and cell regulation: role as a suppressor protein in signal transduction.Mol Cell Biochem. 2011 Jul;353(1-2):101-37. doi: 10.1007/s11010-011-0779-4. Epub 2011 Mar 24. Mol Cell Biochem. 2011. PMID: 21431902 Review.
Cited by
-
Logistic PCA explains differences between genome-scale metabolic models in terms of metabolic pathways.PLoS Comput Biol. 2024 Jun 24;20(6):e1012236. doi: 10.1371/journal.pcbi.1012236. eCollection 2024 Jun. PLoS Comput Biol. 2024. PMID: 38913731 Free PMC article.
-
Extracellular Regucalcin: A Potent Suppressor in the Cancer Cell Microenvironment.Cancers (Basel). 2025 Jan 13;17(2):240. doi: 10.3390/cancers17020240. Cancers (Basel). 2025. PMID: 39858022 Free PMC article. Review.
-
Exploring the Molecular Tapestry: Organ-Specific Peptide and Protein Ultrafiltrates and Their Role in Therapeutics.Int J Mol Sci. 2024 Mar 1;25(5):2863. doi: 10.3390/ijms25052863. Int J Mol Sci. 2024. PMID: 38474110 Free PMC article.
-
Downregulated Regucalcin Expression Induces a Cancer-like Phenotype in Non-Neoplastic Prostate Cells and Augments the Aggressiveness of Prostate Cancer Cells: Interplay with the G Protein-Coupled Oestrogen Receptor?Cancers (Basel). 2024 Nov 24;16(23):3932. doi: 10.3390/cancers16233932. Cancers (Basel). 2024. PMID: 39682121 Free PMC article.
References
-
- Elies J., Yanez M., Pereira T.M.C., Gil-Longo J., MacDougall D.A., Campos-Toimil M. An update to calcium binding proteins. Adv. Exp. Med. Biol. 2020;1131:183–213. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

