Molecular Cloning, Heterologous Expression, Purification, and Evaluation of Protein-Ligand Interactions of CYP51 of Candida krusei Azole-Resistant Fungal Strain
- PMID: 38001874
- PMCID: PMC10668980
- DOI: 10.3390/biomedicines11112873
Molecular Cloning, Heterologous Expression, Purification, and Evaluation of Protein-Ligand Interactions of CYP51 of Candida krusei Azole-Resistant Fungal Strain
Abstract
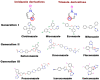
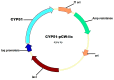
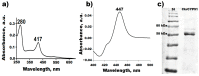
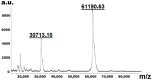
Due to the increasing prevalence of fungal diseases caused by fungi of the genus Candida and the development of pathogen resistance to available drugs, the need to find new effective antifungal agents has increased. Azole antifungals, which are inhibitors of sterol-14α-demethylase or CYP51, have been widely used in the treatment of fungal infections over the past two decades. Of special interest is the study of C. krusei CYP51, since this fungus exhibit resistance not only to azoles, but also to other antifungal drugs and there is no available information about the ligand-binding properties of CYP51 of this pathogen. We expressed recombinant C. krusei CYP51 in E. coli cells and obtained a highly purified protein. Application of the method of spectrophotometric titration allowed us to study the interaction of C. krusei CYP51 with various ligands. In the present work, the interaction of C. krusei CYP51 with azole inhibitors, and natural and synthesized steroid derivatives was evaluated. The obtained data indicate that the resistance of C. krusei to azoles is not due to the structural features of CYP51 of this microorganism, but rather to another mechanism. Promising ligands that demonstrated sufficiently strong binding in the micromolar range to C. krusei CYP51 were identified, including compounds 99 (Kd = 1.02 ± 0.14 µM) and Ch-4 (Kd = 6.95 ± 0.80 µM). The revealed structural features of the interaction of ligands with the active site of C. krusei CYP51 can be taken into account in the further development of new selective modulators of the activity of this enzyme.
Keywords: CYP51; Candida krusei; antifungal drugs; azole inhibitors; cytochrome P450; drug resistance; heterocyclic analogues of steroids; lanosterol 14-alpha demethylase; marine steroids.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Structural analyses of Candida albicans sterol 14α-demethylase complexed with azole drugs address the molecular basis of azole-mediated inhibition of fungal sterol biosynthesis.J Biol Chem. 2017 Apr 21;292(16):6728-6743. doi: 10.1074/jbc.M117.778308. Epub 2017 Mar 3. J Biol Chem. 2017. PMID: 28258218 Free PMC article.
-
Characteristics of the heterologously expressed human lanosterol 14alpha-demethylase (other names: P45014DM, CYP51, P45051) and inhibition of the purified human and Candida albicans CYP51 with azole antifungal agents.Yeast. 1999 Jun 30;15(9):755-63. doi: 10.1002/(SICI)1097-0061(19990630)15:9<755::AID-YEA417>3.0.CO;2-8. Yeast. 1999. PMID: 10398344
-
Impact of Homologous Resistance Mutations from Pathogenic Yeast on Saccharomyces cerevisiae Lanosterol 14α-Demethylase.Antimicrob Agents Chemother. 2018 Feb 23;62(3):e02242-17. doi: 10.1128/AAC.02242-17. Print 2018 Mar. Antimicrob Agents Chemother. 2018. PMID: 29263059 Free PMC article.
-
Recent advances in antifungal drug development targeting lanosterol 14α-demethylase (CYP51): A comprehensive review with structural and molecular insights.Chem Biol Drug Des. 2023 Sep;102(3):606-639. doi: 10.1111/cbdd.14266. Epub 2023 May 23. Chem Biol Drug Des. 2023. PMID: 37220949 Review.
-
Fungal cytochrome P450 sterol 14α-demethylase (CYP51) and azole resistance in plant and human pathogens.Appl Microbiol Biotechnol. 2012 Aug;95(4):825-40. doi: 10.1007/s00253-012-4195-9. Epub 2012 Jun 12. Appl Microbiol Biotechnol. 2012. PMID: 22684327 Review.
Cited by
-
Endophytic strategies decoded by genome and transcriptome analysis of Fusarium nematophilum strain NQ8GII4.Front Microbiol. 2025 Jan 15;15:1487022. doi: 10.3389/fmicb.2024.1487022. eCollection 2024. Front Microbiol. 2025. PMID: 39881987 Free PMC article.
References
-
- Bouopda Tamo S.P. Candida Infections: Clinical Features, Diagnosis and Treatment. Infect. Dis. Clin. Microbiol. 2020;2:91–102. doi: 10.36519/idcm.2020.0006. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

