Inhibiting CBP Decreases AR Expression and Inhibits Proliferation in Benign Prostate Epithelial Cells
- PMID: 38002029
- PMCID: PMC10669082
- DOI: 10.3390/biomedicines11113028
Inhibiting CBP Decreases AR Expression and Inhibits Proliferation in Benign Prostate Epithelial Cells
Abstract
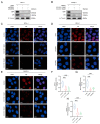
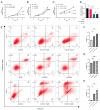
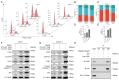
(1) Background: CREB-binding protein (CBP) is a key transcriptional coactivator of androgen receptors (AR). We conducted this study to investigate the effects of CBP on AR expression and proliferation in benign prostatic hyperplasia (BPH) prostate epithelial cells. (2) Methods: By analyzing a published data set, we found that CBP was closely related to the gene expression of AR in prostate cells. We enrolled 20 BPH patients who underwent transurethral resection of the prostate (TURP) in Peking University First Hospital in 2022, and analyzed the expressions of CBP and AR in BPH prostate tissues. Then, we used ICG-001 and shRNA to inhibit CBP in prostate epithelial cells (BPH-1 cells and RWPE-1 cells), and conducted immunofluorescence, cell viability assay, flow cytometry analysis, and Western blot to analyze the effects of CBP on AR expression and proliferation in prostate epithelial cells. We also studied the interaction between CBP and AR through a co-immunoprecipitation assay. (3) Results: CBP is consistent with AR in expression intensity in prostate tissues. Inhibiting CBP decreases AR expression, and induces proliferation inhibition, apoptosis, and cell cycle arrest in BPH prostate epithelial cells. The co-immunoprecipitation assay showed that CBP binds with AR to form transcription complexes in prostate epithelial cells. (4) Conclusions: Inhibiting CBP decreases AR expression and inhibits proliferation in benign prostate epithelial cells. CBP may be a potential target to affect AR expression and the proliferation of prostate epithelial cells in BPH.
Keywords: CBP; androgen receptor; benign prostatic hyperplasia.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Sex steroid receptor expression and localization in benign prostatic hyperplasia varies with tissue compartment.Differentiation. 2013 Apr-Jun;85(4-5):140-9. doi: 10.1016/j.diff.2013.02.006. Epub 2013 Jun 20. Differentiation. 2013. PMID: 23792768 Free PMC article.
-
Macrophages affect immune inflammation and proliferation in benign prostatic hyperplasia via androgen receptor and CD40/CD40L signaling pathway.Tissue Cell. 2020 Jun;64:101343. doi: 10.1016/j.tice.2020.101343. Epub 2020 Jan 23. Tissue Cell. 2020. PMID: 32473708
-
Targeting androgen receptor to suppress macrophage-induced EMT and benign prostatic hyperplasia (BPH) development.Mol Endocrinol. 2012 Oct;26(10):1707-15. doi: 10.1210/me.2012-1079. Epub 2012 Aug 21. Mol Endocrinol. 2012. PMID: 22915828 Free PMC article.
-
Androgen receptor roles in the development of benign prostate hyperplasia.Am J Pathol. 2013 Jun;182(6):1942-9. doi: 10.1016/j.ajpath.2013.02.028. Epub 2013 Apr 6. Am J Pathol. 2013. PMID: 23570837 Free PMC article. Review.
-
The role of the androgen receptor in prostate development and benign prostatic hyperplasia: A review.Asian J Urol. 2020 Jul;7(3):191-202. doi: 10.1016/j.ajur.2019.10.003. Epub 2019 Oct 19. Asian J Urol. 2020. PMID: 32742923 Free PMC article. Review.
References
-
- Fu X., Liu J., Liu D., Zhou Y., Guo Y., Wang Z., Yang S., He W., Chen P., Wang X., et al. Glucose-Regulated Protein 78 Modulates Cell Growth, Epithelial-Mesenchymal Transition, and Oxidative Stress in the Hyperplastic Prostate. Cell Death Dis. 2022;13:78. doi: 10.1038/s41419-022-04522-4. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

