CD39+CD55- Fb Subset Exhibits Myofibroblast-Like Phenotype and Is Associated with Pain in Osteoarthritis of the Knee
- PMID: 38002046
- PMCID: PMC10669511
- DOI: 10.3390/biomedicines11113047
CD39+CD55- Fb Subset Exhibits Myofibroblast-Like Phenotype and Is Associated with Pain in Osteoarthritis of the Knee
Abstract
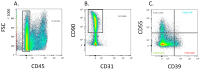
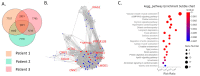
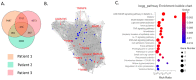
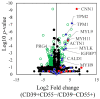
Recent studies utilizing single-cell analysis have unveiled the presence of various fibroblast (Fb) subsets within the synovium under inflammatory conditions in osteoarthritis (OA), distinguishing them from those in rheumatoid arthritis (RA). Moreover, it has been reported that pain in knee OA patients is linked to specific fibroblast subsets. Single-cell expression profiling methods offer an incredibly detailed view of the molecular states of individual cells. However, one limitation of these methods is that they require the destruction of cells during the analysis process, rendering it impossible to directly assess cell function. In our study, we employ flow cytometric analysis, utilizing cell surface markers CD39 and CD55, in an attempt to isolate fibroblast subsets and investigate their relationship with OA pathology. Synovial tissues were obtained from 25 knee OA (KOA) patients. Of these, six samples were analyzed by RNA-seq (n = 3) and LC/MS analysis (n = 3). All 25 samples were analyzed to estimate the proportion of Fb (CD45-CD31-CD90+) subset by flow cytometry. The proportion of Fb subsets (CD39+CD55- and CD39-CD55+) and their association with osteoarthritis pathology were evaluated. CD39+CD55- Fb highly expressed myogenic markers such as CNN1, IGFBP7, MYH11, and TPM1 compared to CD39-CD55+ Fb. Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis of upregulated differentially expressed genes (DEGs) in CD39+CD55- Fb identified the Apelin pathway and cGMP-PKC-signaling pathway as possibly contributing to pain. LC/MS analysis indicated that proteins encoded by myogenic marker genes, including CNN1, IGFBP7, and MYH11, were also significantly higher than in CD39-CD55+ Fb. CD39-CD55+ Fb highly expressed PRG4 genes and proteins. Upregulated DEGs were enriched for pathways associated with proinflammatory states ('RA', 'TNF signaling pathway', 'IL-17 signaling pathway'). The proportion of CD39+CD55- Fb in synovium significantly correlated with both resting and active pain levels in knee OA (KOA) patients (resting pain, ρ = 0.513, p = 0.009; active pain, ρ = 0.483, p = 0.015). There was no correlation between joint space width (JSW) and the proportion of CD39+CD55- Fb. In contrast, there was no correlation between the proportion of CD39-CD55+ Fb and resting pain, active pain, or JSW. In conclusion, CD39+CD55- cells exhibit a myofibroblast phenotype, and its proportion is associated with KOA pain. Our study sheds light on the potential significance of CD39+CD55- synovial fibroblasts in osteoarthritis, their myofibroblast-like phenotype, and their association with joint pain. These findings provide a foundation for further research into the mechanisms underlying fibrosis, the impact of altered gene expression on osteoarthritic joints, and potential therapeutic strategies.
Keywords: CD39; fibroblast subset; knee osteoarthritis; pain.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
IL24 Expression in Synovial Myofibroblasts: Implications for Female Osteoarthritis Pain through Propensity Score Matching Analysis.Medicina (Kaunas). 2024 Apr 29;60(5):741. doi: 10.3390/medicina60050741. Medicina (Kaunas). 2024. PMID: 38792924 Free PMC article.
-
Fibrocyte Phenotype of ENTPD1+CD55+ Cells and Its Association with Pain in Osteoarthritic Synovium.Int J Mol Sci. 2024 Apr 6;25(7):4085. doi: 10.3390/ijms25074085. Int J Mol Sci. 2024. PMID: 38612896 Free PMC article.
-
Identification and validation of hub genes of synovial tissue for patients with osteoarthritis and rheumatoid arthritis.Hereditas. 2021 Sep 28;158(1):37. doi: 10.1186/s41065-021-00201-0. Hereditas. 2021. PMID: 34583778 Free PMC article.
-
Identification of HBEGF+ fibroblasts in the remission of rheumatoid arthritis by integrating single-cell RNA sequencing datasets and bulk RNA sequencing datasets.Arthritis Res Ther. 2022 Sep 6;24(1):215. doi: 10.1186/s13075-022-02902-x. Arthritis Res Ther. 2022. PMID: 36068607 Free PMC article.
-
Comparison of cathepsins K and S expression within the rheumatoid and osteoarthritic synovium.Arthritis Rheum. 2002 Mar;46(3):663-74. doi: 10.1002/art.10114. Arthritis Rheum. 2002. PMID: 11920402
Cited by
-
Association Between Synovial NTN4 Expression and Pain Scores, and Its Effects on Fibroblasts and Sensory Neurons in End-Stage Knee Osteoarthritis.Cells. 2025 Mar 8;14(6):395. doi: 10.3390/cells14060395. Cells. 2025. PMID: 40136644 Free PMC article.
-
Shotgun Proteomics Links Proteoglycan-4+ Extracellular Vesicles to Cognitive Protection in Amyotrophic Lateral Sclerosis.Biomolecules. 2024 Jun 19;14(6):727. doi: 10.3390/biom14060727. Biomolecules. 2024. PMID: 38927130 Free PMC article.
-
IL24 Expression in Synovial Myofibroblasts: Implications for Female Osteoarthritis Pain through Propensity Score Matching Analysis.Medicina (Kaunas). 2024 Apr 29;60(5):741. doi: 10.3390/medicina60050741. Medicina (Kaunas). 2024. PMID: 38792924 Free PMC article.
-
Current cutting-edge omics techniques on musculoskeletal tissues and diseases.Bone Res. 2025 Jun 9;13(1):59. doi: 10.1038/s41413-025-00442-z. Bone Res. 2025. PMID: 40484858 Free PMC article. Review.
-
Fibrocyte Phenotype of ENTPD1+CD55+ Cells and Its Association with Pain in Osteoarthritic Synovium.Int J Mol Sci. 2024 Apr 6;25(7):4085. doi: 10.3390/ijms25074085. Int J Mol Sci. 2024. PMID: 38612896 Free PMC article.
References
-
- Nanus D.E., Wijesinghe S.N., Pearson M.J., Hadjicharalambous M.R., Rosser A., Davis E.T., Lindsay M.A., Jones S.W. Regulation of the Inflammatory Synovial Fibroblast Phenotype by Metastasis-Associated Lung Adenocarcinoma Transcript 1 Long Noncoding RNA in Obese Patients with Osteoarthritis. Arthritis Rheumatol. 2020;72:609–619. doi: 10.1002/art.41158. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

