The Innovative Biomaterials and Technologies for Developing Corneal Endothelium Tissue Engineering Scaffolds: A Review and Prospect
- PMID: 38002407
- PMCID: PMC10669703
- DOI: 10.3390/bioengineering10111284
The Innovative Biomaterials and Technologies for Developing Corneal Endothelium Tissue Engineering Scaffolds: A Review and Prospect
Abstract
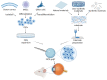
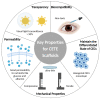
Corneal transplantation is the only treatment for corneal endothelial blindness. However, there is an urgent need to find substitutes for corneal endothelium grafts due to the global shortage of donor corneas. An emerging research field focuses on the construction of scaffold-based corneal endothelium tissue engineering (CETE). Long-term success in CETE transplantation may be achieved by selecting the appropriate biomaterials as scaffolds of corneal endothelial cells and adding bioactive materials to promote cell activity. This article reviews the research progress of CETE biomaterials in the past 20 years, describes the key characteristics required for corneal endothelial scaffolds, and summarizes the types of materials that have been reported. Based on these, we list feasible improvement strategies for biomaterials innovation. In addition, we describe the improved techniques for the scaffolds' surface topography and drug delivery system. Some promising technologies for constructing CETE are proposed. However, some questions have not been answered yet, and clinical trials and industrialization should be carried out with caution.
Keywords: biomaterials; corneal endothelium tissue engineering; drug delivery; innovative technologies; surface topography.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Advancements in Polymer Biomaterials as Scaffolds for Corneal Endothelium Tissue Engineering.Polymers (Basel). 2024 Oct 12;16(20):2882. doi: 10.3390/polym16202882. Polymers (Basel). 2024. PMID: 39458711 Free PMC article. Review.
-
Corneal endothelium tissue engineering: An evolution of signaling molecules, cells, and scaffolds toward 3D bioprinting and cell sheets.J Cell Physiol. 2020 Oct 8. doi: 10.1002/jcp.30085. Online ahead of print. J Cell Physiol. 2020. PMID: 33090510 Review.
-
Research Progress of Polymer Biomaterials as Scaffolds for Corneal Endothelium Tissue Engineering.Nanomaterials (Basel). 2023 Jun 29;13(13):1976. doi: 10.3390/nano13131976. Nanomaterials (Basel). 2023. PMID: 37446492 Free PMC article. Review.
-
Applications of Biomaterials in Corneal Endothelial Tissue Engineering.Cornea. 2016 Nov;35 Suppl 1:S25-S30. doi: 10.1097/ICO.0000000000000992. Cornea. 2016. PMID: 27617875 Review.
-
The Human Tissue-Engineered Cornea (hTEC): Recent Progress.Int J Mol Sci. 2021 Jan 28;22(3):1291. doi: 10.3390/ijms22031291. Int J Mol Sci. 2021. PMID: 33525484 Free PMC article. Review.
Cited by
-
Innovative Bioscaffolds in Stem Cell and Regenerative Therapies for Corneal Pathologies.Bioengineering (Basel). 2024 Aug 23;11(9):859. doi: 10.3390/bioengineering11090859. Bioengineering (Basel). 2024. PMID: 39329601 Free PMC article. Review.
-
Advancements in Polymer Biomaterials as Scaffolds for Corneal Endothelium Tissue Engineering.Polymers (Basel). 2024 Oct 12;16(20):2882. doi: 10.3390/polym16202882. Polymers (Basel). 2024. PMID: 39458711 Free PMC article. Review.
-
Focus on seed cells: stem cells in 3D bioprinting of corneal grafts.Front Bioeng Biotechnol. 2024 Jul 10;12:1423864. doi: 10.3389/fbioe.2024.1423864. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 39050685 Free PMC article. Review.
-
Comparison of cellular, mechanical, and optical properties of different polymers for corneal tissue engineering.Iran J Basic Med Sci. 2025;28(8):1082-1099. doi: 10.22038/ijbms.2025.85468.18477. Iran J Basic Med Sci. 2025. PMID: 40584436 Free PMC article.
-
Advancements in bioengineering for descemet membrane endothelial keratoplasty (DMEK).NPJ Regen Med. 2025 Feb 14;10(1):10. doi: 10.1038/s41536-025-00396-0. NPJ Regen Med. 2025. PMID: 39952985 Free PMC article. Review.
References
-
- Hos D., Matthaei M., Bock F., Maruyama K., Notara M., Clahsen T., Hou Y., Le V.N.H., Salabarria A.-C., Horstmann J., et al. Immune Reactions after Modern Lamellar (DALK, DSAEK, DMEK) versus Conventional Penetrating Corneal Transplantation. Prog. Retin. Eye Res. 2019;73:100768. doi: 10.1016/j.preteyeres.2019.07.001. - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

